SavageKidKobe
21.07.2019 •
Chemistry
The concentration of the stock solution she needs is 100 milli molar (mm) and she needs to make 1.2 milli liters (ml) solution of the drug-a. the drug is available in a salt form with a molecular weight of 181.6 grams / mole. what is the amount (quantity in grams) of drug-a will she have to weigh-out in order to make the stock solution
Solved
Show answers
More tips
- H Health and Medicine Why Do Humans Have One Heart?...
- S Style and Beauty How to knit a hooded cowl?...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- S Style and Beauty Tricks and Tips: How to Get Rid of Freckles...
- H Health and Medicine What is Autism? Understanding the Basics of This Neurodevelopmental Disorder...
- F Family and Home How to Properly Use a Water Level?...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
Answers on questions: Chemistry
- H History HELEPPOP which amendment has been very controversial in modern america; but has been greatly backed by conservatives who believe in their right to protect themselves...
- B Biology What is photosynthesis...
- M Mathematics Each equation represents a portions relationship. Choose the equations for which the constant of proportionality is 3/4...

Ответ:
molarity of stock solution to be prepared - 100 x 10⁻³ mol/L
volume of stock solution to be prepared - 1.2 mL
Therefore number of moles in 1.2 mL - 100 x 10⁻³ mol/L x 1.2 x 10⁻³ L
number of moles of drug - 1.2 x 10⁻⁴ mol
mass of drug required - 1.2 x 10⁻⁴ mol x 181.6 g/mol = 21. 8 mg
21.8 g of drug is required to make the stock solution
Ответ:
Answer : The volume of in Reaction 1 = 240.45 ml
in Reaction 1 = 240.45 ml
The volume of in Reaction 2 = 480.89 ml
in Reaction 2 = 480.89 ml
Solution : Given,
Temperature = (
(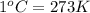 )
)
Pressure = 0.900 atm
The mass of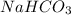 = 1 gram
= 1 gram
Molar mass of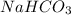 = 84.007 g/mole
= 84.007 g/mole
The given reactions are,
Reaction 1 :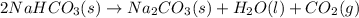
Reaction 2 :
Calculation for Reaction 1 :First we have to calculate the moles of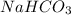 .
.
Moles of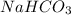 =
= 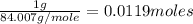
From the Reaction 1, we conclude that
2 moles of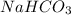 gives 1 mole of
gives 1 mole of 
0.0119 moles of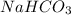 gives
gives  of
of 
Using ideal gas equation :
where,
P = pressure of gas
V = volume of gas
n = Number of moles
T = temperature of gas
R = gas constant = 0.0821 L atm/mole K
Now put all the given values in ideal gas law, we get the volume of
The volume of in Reaction 1 is 240.45 ml
in Reaction 1 is 240.45 ml
Calculation for Reaction 2 :The moles of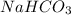 = 0.0119 moles
= 0.0119 moles
From the Reaction 2, we conclude that
1 moles of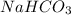 gives 1 mole of
gives 1 mole of 
So, the moles of = the moles of
= the moles of 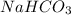 = 0.0119 moles
= 0.0119 moles
Using ideal gas equation,
Now put all the given values in ideal gas law, we get the volume of
The volume of in Reaction 2 is 480.89 ml
in Reaction 2 is 480.89 ml