haleyehewitt2001
30.03.2020 •
Chemistry
The decay constant for 14C is .00012 In 1947, the famous cave paintings in Lascaux, France were discovered and testing revealed that charcoal in the cave contained 20% of the 14C found in living trees. Write a formula for the age of the charcoal (hence of the associated paintings). Show your work to find this formula.
Solved
Show answers
More tips
- C Computers and Internet What is Web 2.0 and How Does it Work?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...

Ответ:
The question is incomplete, here is the complete question:
The decay constant for 14-C is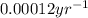 In 1947, the famous cave paintings in Lascaux, France were discovered and testing revealed that charcoal in the cave contained 20% of the 14-C found in living trees. Write a formula for the age of the charcoal (hence of the associated paintings). Show your work to find this formula.
In 1947, the famous cave paintings in Lascaux, France were discovered and testing revealed that charcoal in the cave contained 20% of the 14-C found in living trees. Write a formula for the age of the charcoal (hence of the associated paintings). Show your work to find this formula.
The formula for the age of the charcoal is
Explanation:
Carbon-14 isotope is a radioisotope and its decay process follows first order kinetics.
Rate law expression for first order kinetics is given by the equation:
where,
k = rate constant =
t = time taken for decay process = ? yr
[A] = amount left after decay process = (100 - 20) = 80 grams
Putting values in above equation, we get:
Hence, the formula for the age of the charcoal is
Ответ:
Not sure sorry
Explanation: