The density of potassium, which has the BCC structure, is 0.855 g/cm3. The atomic weight of potassium is 39.09 g/mol. Calculate (a) the lattice parameter; and (b) the atomic radius of potassium.
Solved
Show answers
More tips
- A Animals and plants 10 Tips for Growing Delicious and High-Quality Tomatoes in Your Garden...
- H Health and Medicine Tick Traps: How to Remove Them Safely and Effectively...
- F Family and Home How to Keep Your Home Warm: Tips and Tricks...
- C Computers and Internet How Much Does an iPhone Cost in America?...
- S Style and Beauty Discover the Art of Nail Design: How Do You Paint Your Nails?...
- F Food and Cooking Discover How to Properly Prepare Dough for Rasstegai...
- F Family and Home Ways to Attract Wealth into Your Home...
- G Goods and services Stock center - a modern way of optimizing logistics...
- F Family and Home How to Teach Your Child to Read?...
- F Food and Cooking How to properly cook shrimp: tips from professionals...
Answers on questions: Chemistry
- C Chemistry If 0.484J of heat is added to 0.1372g of water, how much will the temperature increase? 0.263 °C 0.927 °c 0.843 °C 0.460 °C...
- C Chemistry The indicator methyl red has different molecular structures at high and low pH. a. True b. False...
- C Chemistry What types of environmental issues are facing the residents of Baltimore? Be specific. Examples of environmental issues include those related to water supply (including...
- H History Do you know this for social studies...
- M Mathematics If the area of a right angle triangle with base 3 cm is 6cm find the height...
- M Mathematics Anyone plz plz help only 4 questions and I will mark as brainliest plz more points...
- H Health 1.4 Describe how the following concepts could help to fight social challenges social and environmental responsibilitysocial activismsocial justice量...
- M Mathematics I really need help because I’m so confused where to start and it’s due tomorrow :(...
- B Biology 13. Determine the mean for each data group, and describe in a complete sentence how you calculated them...
- M Mathematics Use the diagram at the right. How many places contain each line and point?...

Ответ:
For a: The edge length of the crystal is 533.5 pm
For b: The atomic radius of potassium is 231.01 pm
Explanation:
For a:To calculate the lattice parameter or edge length of the crystal, we use the equation:
where,
Z = number of atom in unit cell = 2 (BCC)
M = atomic mass of metal = 39.09 g/mol
a = edge length of unit cell = ?
Putting values in above equation, we get:
Conversion factor: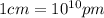
Hence, the edge length of the crystal is 533.5 pm
For b:To calculate the edge length, we use the relation between the radius and edge length for BCC lattice:
where,
R = radius of the lattice = ?
a = edge length = 533.5 pm
Putting values in above equation, we get:
Hence, the atomic radius of potassium is 231.01 pm
Ответ:
15 feet by 12 feet or 10 feet by 18 feet
Step-by-step explanation: