The enthalpy of vaporization of liquid water is 40.65 kJ/mol. Calculate the energy required to vaporize 12.5 g of liquid water.
Solved
Show answers
More tips
- C Computers and Internet How to Download Movies from Torrents?...
- F Food and Cooking How to Make the Perfect Glühwein: Step-by-Step Guide...
- A Animals and plants How to Grow Lime from a Seed: Simple Tips and Interesting Facts...
- S Style and Beauty How to Properly Tie a Tie: 5 Simple Steps...
- C Computers and Internet Dynamically Assigned IP Address: What Is It and How Does It Work?...
- C Computers and Internet How to Check the Speed of My Internet?...
- H Health and Medicine 5 Simple Steps to Quit Smoking for Good...
- C Computers and Internet How to Download Videos from YouTube? Simple Steps to Download Any Content...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- S Style and Beauty How to Get Rid of Acne: Scientifically Proven Methods...
Answers on questions: Chemistry
- C Chemistry What are the names of the compound (with roman numeral)? Na2SO4...
- C Chemistry 9.25x10^-8 x6.40x10^3 in scientific equation...
- C Chemistry 1. In which area are you most likely to find plants that can tolerate water that has a constantly changing salt content? A. a freshwater stream B. an estuary C. a a pond D. a coral...
- C Chemistry Question 2 of 25 How can a reaction generally be made to go more quickly? Check all that apply. O A. Cooling the reaction O B. Lowering the activation energy O c. Adding a catalyst...
- C Chemistry Please please please please please please help me a b c or d...
- C Chemistry Match the atoms which might substitute for one another. 1. Si V 2. K As 3. Nb Ca 4. Cu Na 5. P C 6. Mg Ag...
- C Chemistry 4NH3(g) + 6NO(g)→5N2(g) + 6H2O(g) Using the balanced equation calculate the mass of N2 produced from 100 grams of NH3 racing with an unlimited supply of NO....
- M Mathematics The lowest recorded temperature in Alaska is -67.8 °C. The highest recorded temperature in Alaska is 43.2 °C. Find the difference between these temperatures....
- E English Identify the productions of the phrase-structure grammar for each of these languages, where S is the starting symbol....
- B Biology A current theory of amenorrhea in athletes is that the disruption of menstruation is a direct result of the disruption of the normal secretion of...

Ответ:
The energy required to vaporize 12.5 g of liquid water is 28.2 kJ
Explanation:
Latent heat of vaporization is the amount of heat required to convert 1 gram of liquid into its vapor state without change in its temperature.
Given : The enthalpy of vaporization of water is 40.65 kJ/mol.
n = number of moles =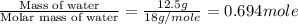
Thus 1 mole of water requires heat = 40.65 kJ
0.694 moles of water requires heat =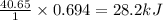
Thus the energy required to vaporize 12.5 g of liquid water is 28.2 kJ
Ответ: