The equilibrium constant of a system, K, can be related to the standard free energy change, ΔG∘, using the following equation: ΔG∘=−RTlnK where T is a specified temperature in kelvins (usually 298 K) and R is equal to 8.314 J/(K⋅mol)Acetylene, C2H2, can be converted to ethane, C2H6, by a process known as hydrogenation. The reaction is C2H2(g)+2H2(g)⇌C2H6(g) Given the following data at standard conditions (all pressures equal to 1 atm and the common reference temperature 298 K), what is the value of Kp for this reaction?
Solved
Show answers
More tips
- C Computers and Internet Why is Yandex s robot Bless.yandex.ru Important?...
- S Science and Technology How to Restore the Diamond Shine to Your Tarnished Silverware...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- L Leisure and Entertainment How to Find a Phone Number by Address: The Ultimate Guide...
- F Food and Cooking Pu-erh Tea: History, Varieties, Benefits, and Risks...
- S Style and Beauty Why is Sugaring Better than Waxing for Hair Removal?...
- C Computers and Internet How to Delete a Page on VKontakte: The Ultimate Guide from Experts...
- F Food and Cooking How to Make Delicious Plov: Secrets and Recipes...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- L Leisure and Entertainment Unlocking the Secrets of Winning the Lottery...
Answers on questions: Chemistry
- C Chemistry Calculate the equivalent weight of Ca(OH)2 in the reaction. HCl + Ca(OH)2 → Ca(OH)Cl + H2O...
- C Chemistry Can someone please help me with this...
- C Chemistry 9. A tank truck carries 34 000 L of sulfuric acid. The density of sulfuric acid is 1.84 kg/L. TAI (a) What mass of sulfuric acid is in the truck?...
- C Chemistry study the diagram and answer the following questions. a. Name A and B. b. Which element is it? Write its name and B symbol. p*= 10 n°= 10 - A C. What is its atomic mass...
- C Chemistry Consider a transition in which the hydrogen atom is excited from n=1 to n=[infinity]. What is the end result of this transition?...
- C Chemistry Calculate the theoretical yield of C2H5Cl when 111 g of C2H6 reacts with 203 g of Cl2, assuming that C2H6 and Cl2 react only to form C2H5Cl and HCl....
- C Chemistry A. Explain why safety glasses CANNOT be worn in lab. Explain why goggles must be worn, instead. b. Explain why long pants and closed-toe/ closed heel shoes must be worn...
- C Chemistry State the Structure of sulfur and phosphorus shown by crystals...
- C Chemistry If the half-life of a radionulide is 1 month, what percent remains after 4 months?...
- C Chemistry Kim dissolves 70.13 g of solid sodium chloride (NaCl) in enough distilled water to make 400 mL of stock solution. What volumes of stock solution and distilled water...

Ответ:
7.98 grams of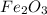 are produced if 10.7 grams of
are produced if 10.7 grams of 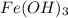 are reacted.
are reacted.
Explanation:
To calculate the number of moles, we use the equation:
Putting values in equation 1, we get:
The chemical equation for the reaction is
By Stoichiometry of the reaction:
2 moles of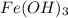 produce = 1 mole of
produce = 1 mole of 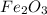
So, 0.100 moles of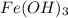 produce=
produce= 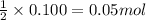 of
of 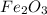
Mass of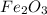 =
=
Hence 7.98 grams of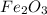 are produced if 10.7 grams of
are produced if 10.7 grams of 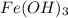 are reacted.
are reacted.