Superstar7180
01.12.2020 •
Chemistry
The image represents a(n).
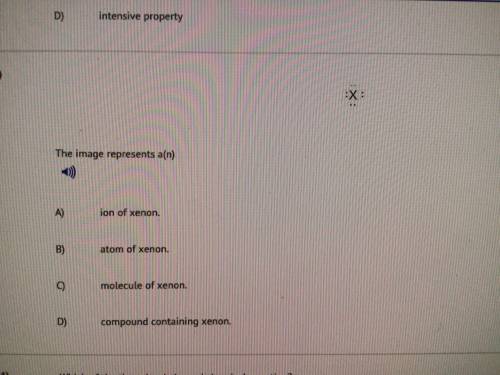
Solved
Show answers
More tips
- B Business and Finance Understanding Cash Flow: What It Is and How It Works...
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...

Ответ:
Balanced chemical reaction: 2H₂ + O₂ → 2H₂O.
n(O₂) = 2.50 mol.
From chemical reaction: n(O₂) : n(H₂O) = 1 : 2.
n(H₂O) = 2 · 2.50 mol.
n(H₂O) = 5.00 mol; amount of substance.
M(H₂O) = 2 · 1 g/mol + 16 g/mol.
M(H₂O) = 18 g/mol; molar mass.
m(H₂O) = n(H₂O) · M(H₂O).
m(H₂O) = 5 mol · 18 g/mol.
m(H₂O) = 90 g; mass of substance.