coralaguilar1702
27.07.2019 •
Chemistry
The ka values for several weak acids are given below. which acid (and its conjugate base) would be the best buffer at ph = 8.0?
Solved
Show answers
More tips
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Calculate the mass of water of hydration if a hydrate loses 0.025 mol of H O when heated....
- C Chemistry Which are accurate descriptions of comparative anatomy? Select three options. may or may not include studies of structures with the same function only involves studies...
- C Chemistry Cells/Prokaryotic vs Eukaryotic (6.12A & B) Question 6 The diagram below BEST represents which part of the cell theory? Select one: 1.all living things are made...
- C Chemistry What are the two main sources of hydrocarbons?...
- C Chemistry What are the values of the solute amount in moles and the solution volume in liters....
- C Chemistry C. rubidium sulfate 2. Write balanced molecular equation, complete ionic equation, and net ionic equations for the mixing of the following solutions. Show states....
- B Business Which of the following would not be on the statement of cash flows? a.cash flows from investing activities b.cash flows from contingent activities c.cash flows from...
- E English Which sentences contain an adverb clause?...
- M Mathematics How many are 3 raised to 5 ???...
- E English We have eaten all of our dinner and our homework is finished. is it parallel or no?...

Ответ:
Tris and its conjugate base would be the best buffer at pH 8.
Further Explanation:
Buffer solution:
The solutions that oppose any change in their pH on addition of small amounts of acid or base are called buffer solutions. These are formed wither by a weak base and its conjugate acid or a weak acid and its conjugate base.
Henderson-Hasselbalch equation for a buffer solution is as follows:
A buffer is most effective when the concentration of both acid and its conjugate base is the same. So equation (1) then modifies as follows:
Therefore a buffer works best if its pH becomes equal to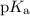 .
.
The formula to calculate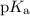 is as follows:
is as follows:
Here,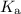 is the dissociation constant of acid.
is the dissociation constant of acid.
Substitute for
for 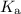 in equation (3) to calculate
in equation (3) to calculate 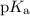 of tris.
of tris.
Substitute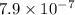 for
for 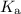 in equation (3) to calculate
in equation (3) to calculate 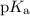 of MES.
of MES.
Substitute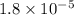 for
for 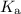 in equation (3) to calculate
in equation (3) to calculate 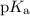 of acetic acid.
of acetic acid.
Substitute for
for 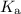 in equation (3) to calculate
in equation (3) to calculate 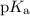 of formic acid.
of formic acid.
Substitute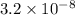 for
for 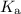 in equation (3) to calculate
in equation (3) to calculate 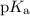 of HEPES.
of HEPES.
The value of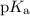 of tris is nearly equal to the required pH. So tris along with its conjugate base would be the best buffer at pH 8.
of tris is nearly equal to the required pH. So tris along with its conjugate base would be the best buffer at pH 8.
Learn more:
Write the chemical equation responsible for pH of buffer containing and :
Reason for the acidic and basic nature of amino acid.
Answer details:
Grade: High School
Chapter: Acid, base and salts
Subject: Chemistry
Keywords: tris, acetic acid, formic acid, MES, HEPES, pH, pKa, Henderson, best buffer, 7.49, 8.20, 4.74, 3.74, Ka.
Ответ: