The kp for the reaction below is 1.49 × 108 at 100.0°c: co(g) + cl2(g) → cocl2(g) in an equilibrium mixture of the three gases, pco = pcl2 = 2.22 × 10-4 atm. the partial pressure of the product, phosgene (cocl2), is atm.
Solved
Show answers
More tips
- S Style and Beauty How to Choose the Perfect Hair Color?...
- C Computers and Internet Best iPad Games: Our Opinion...
- A Animals and plants Man s Best Friend: Which Dog Breed Is the Most Friendly?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry Arrange the compounds by boiling point. a. A pentane molecule with the condensed structure CH3CH2CH2CH2CH3. b. A neopentane molecule with the condensed structure C(CH3)4. c. A...
- C Chemistry Determine the number of significant figures in each of the following measurements:. (a) 0.0000003 cm (b) 2901 g (c) 40.2 g (d) 56 mL...
- C Chemistry Based on your observations how are the top left and bottom left numbers of an atom symbol different?Explain Answer as fast as you can I need it right now please...
- C Chemistry What is the identity of the mystery ink in the note? paper chromatography...
- C Chemistry Which were the first heavy elements to be created in a laboratory, and where and when were they created?...
- C Chemistry A flask that weighs 0.4532 kg is filled with 0.200 L of carbon tetrachloride. The weight of the flask and carbon tetrachloride is found to be 789.6 g. What is the density of the...
- C Chemistry Can anyone help me please...
- C Chemistry What method do all scientists, across all fields, use in the pursuit of scientific knowledge? 1. They generate or use empirical evidence to study something about the natural world....
- C Chemistry In order to scale a recipe, one must...
- C Chemistry Class science A revolution occurs: Question 14 options: Each time the Sun rotates around the Earth Each time the Earth turns once on its axis Each time the Earth orbits the sun...

Ответ:
7.34 atm
Further explanationGiven:
The Kp for the reaction below is 1.49 · 10⁸ at 100.0°C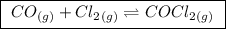 (a balanced reaction)
(a balanced reaction)
In an equilibrium mixture of the three gases.
p CO = p Cl₂ = 2.22 · 10⁻⁴ atm.
Question:
The partial pressure of the product, phosgene (COCl₂), is __ atm.
The Process:
Let us write the equilibrium constant in terms of pressure based on the reaction above.
We set p COCl₂ as the subject to be asked.
Substitute all the data above into the equation.
Thus, the partial pressure of the product, phosgene (COCl₂), is 7.34 atm.
_ _ _ _ _ _ _ _ _ _
Notes:
For the general reactions: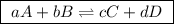 ,
,
the equilibrium constant in terms of concentrations is given by the expression:
![\boxed{ \ K_c = \frac{[C]^c[D^d]}{[A]^a[B]^b} \ }](/tpl/images/0273/3667/d057b.png)
Learn moreWrite the equilibrium constant for the reaction Write the equilibrium constant for the reaction of a heterogeneous balance. What is [S²⁻] at equilibrium?Ответ:
The partial pressure of phosgene is
is 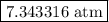 .
.
Further explanation:
Chemical equilibrium:
The state where reactant concentration and product concentration become constant and do not change with time is known as equilibrium.The rate of forward and backward reactions becomes equal at equilibrium.
Equilibrium constant in pressure terms:
The ratio of partial pressures of products to partial pressures of reactants, both of these terms are raised to some power equal to their respective coefficients in a balanced chemical equation. It is denoted by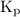 .
.
Consider a general balanced reaction,
The formula to calculate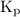 is as follows:
is as follows:
Here,
a and b are stoichiometric coefficients of A and B respectively.
c and d are stoichiometric coefficients of C and D respectively.
Given reaction is as follows:
The expression for equilibrium constant for this reaction is as follows:
Where,
Rearrange equation (1) to calculate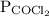 .
.
Substitute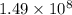 for
for 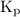 ,
, 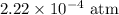 for
for 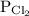 and
and 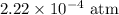 for
for 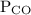 in equation (2).
in equation (2).
Learn more:
Sort the solubility of gas will increase or decrease: . What is the pressure of the gas?: .Answer details:
Grade: Senior School
Subject: Chemistry
Chapter: Chemical equilibrium
Keywords: Kp, CO, Cl2, COCl2, 7.343316 atm, chemical equilibrium, pressure, partial pressure, phosgene, PCl2, PCO, PCOCl2.
Ответ: