The mass of one mole of sugar is 342 g. you used 19 g of sugar. calculate the number of moles of sugar you used.
Solved
Show answers
More tips
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- H Health and Medicine How does childbirth happen: everything future mothers need to know...
- H Health and Medicine What Are the Best Vitamins? A Scientific View on Vitamin Supplements...
- A Auto and Moto How to Deregister a Car in Moscow?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- A Auto and Moto How to choose the right drive for your BMW...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- L Leisure and Entertainment Mysteries of the Name Vyacheslav Dobrynin...
- H Health and Medicine What makes Graves’ disease dangerous?...
- F Food and Cooking When is Easter in 2011?...
Answers on questions: Chemistry
- C Chemistry What initially forms as pale blue and pale green precipitate when nh3 is added to cuh2o6?...
- C Chemistry In the following reaction, when a surplus of hydrazine (n2h4) reacts with 20 liters of hydrogen peroxide (h2o2), how many liters of nitrogen gas will be released,...
- C Chemistry Why does burning wood leaves an ash that is only a small fraction of the mass of the original wood.?...
- C Chemistry When standardizing an iodine solution, 30.37 ml of the solution was required to completely oxidize 10.00 ml of a 5 mg/ml solution of ascorbic acid. what is the molarity...
- C Chemistry A teacher tells you that they have 30 cm3 of sand (volume) in a graduated cylinder. You tell the teacher that this is not accurate since sand has a bunch of air spaces...
- C Chemistry Which statement correctly describes the conductivity of the specified material? Gold has a high conductivity Most plastics have a high conductivity Most wires are...
- M Mathematics The equation y=5.5 represents a proportional relationship what is the constant of proportionally...
- M Mathematics A newspaper article reports the results of an election between two candidates. The article says that Smith received 10% of the votes and that Murphy received 1 2...
- S Social Studies In the chart, name an artifact for each characteristic of American civilization. Then tell what you think the artifact reveals about the characteristic it relates...
- S Social Studies Larry has been recently promoted to the position of a team lead at an insurance company. This promotion was based on his boss s assessment that Larry is capable of...

Ответ:
The number of moles of sugar that is used are 0.055 moles.
Explanation:
We are given:
Mass of 1 mole of sugar = 342 g
Mass of sugar used = 19 g
So, by applying unitary method, we get:
342 grams of sugar is making 1 mole
So, 19 grams of sugar will make =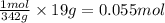
Hence, the number of moles of sugar that is used are 0.055 moles.
Ответ:
1. K increases
2. K doesn't change
3. K decreases
4. K doesn't change
5. K doesn't change
6. K increases
7. K doesn't change
Explanation:
The equilibrium constant for a reaction is always the same value at a given temperature. So, any change in temperature will change K value.
Le chatelier principle states that if a change is imposed on a system at equilibrium, the position of the equilibrium will shift in a direction that tends to reduce that change, then we have:
Increase T: Since it is an endothermic reaction when temperature is raised the position of the chemical equilibrium will lie to the right because the shift will be in the direction that consumes energy. And K will increaseRemove H2 (T constant) will cause the system to shift to the left and H2 increases in that way. K doesn't changeDecrease T: It will decrease K and shift the equilibrium to the left.Decrease the volume (constant T): when the volume is reduced, the system responds by reducing its volume. However, reactants and products have the same number of moles so decreasing volume doesn't affect the equilibrium position nor K value.Add Ne(g): the addition of an inert gas like neon increases the total pressure but does not affect the concentrations of the reactants or products because it doesn't participate in the reaction. K will not change.Add H2 will shift the equilibrium to the right to decrease H2 concentration. K will increase Add a catalyst will increase the rate of reaction but it doesn't affect K value.