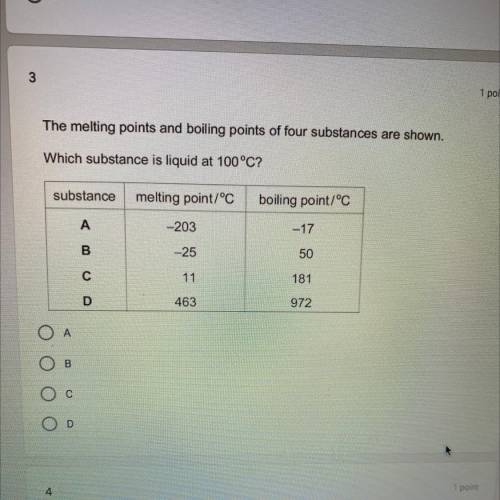
The melting points and boiling points of 4 substances are shown.
which substance is a liquid at 100 C?
A- melting( -203, boil (-17
B- melt (-25, boyl 50
C- melt 11 , boil 181
D- melt 463 boil 972
Solved
Show answers
More tips
- D Dating, Love, Relationships Is it a Compliment or Flattery: What s the Difference?...
- H Health and Medicine What Are the Best Vitamins? A Scientific View on Vitamin Supplements...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- C Computers and Internet How to Set Up Internet on iPhone? Detailed Guide with Step-by-Step Instructions...
- F Family and Home How to Teach Your Child to Speak: Tips and Recommendations...
- P Philosophy Agnosticism: Opinion or Belief?...
- S Style and Beauty How to choose the best mascara for your eyelashes...
- F Food and Cooking Discover Delicious Recipes You Can Make with Ground Meat...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- S Science and Technology Why is there no gravity on other planets?...
Answers on questions: Chemistry
- C Chemistry Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?...
- C Chemistry Why is it important for dna to be duplicated before mitosis...
- C Chemistry What do all four of these types of molecules have in common? a. they contain instructions. b. they are motors for movement. c. each is only involved in an organism...
- C Chemistry [] A constant force of 900 N pushes a 100 kg mass up the inclined plane at a uniform speed of 4 m/s. The work done by the pushing force is... a. 20,000 J b. 1000J...
- C Chemistry What is the volume (liters) of 85.0 grams of F2 gas at STP?*...
- C Chemistry Consider the following chemical reaction: 2SO2 (g) + O2 (g) 2SO3 (g) 1.50 L. of sulfur trioxide at the pressure of 1.20 atm. and temperature of 25 oC is mixed with...
- C Chemistry Arrange the steps in order to describe how photons and carbon dioxide (CO2) contribute to the warming of Earth. CO2 in the atmosphere absorbs infrared photons. CO2...
- C Chemistry Do you think that the human being is the center of the universe?...
- C Chemistry Do you think that people decide their destiny, or that their destiny has already been decided and they cannot change ,they only discover it ?...
- C Chemistry Does seggs hurt? cause some people said it did and some said it dont....

Ответ:
it's
C. with melting point 11° C and boiling point 181° C
Explanation:
i hope you got it
Ответ:
These are four questions, each with its complete answer.
Question 1. If the temperature of a gas remains constant, then the pressure of the gas will increase if the a. mass of the gas molecules decreases. b. diffusion of the gas molecules increases. c. size of the container is decreased. d. number of gas molecules in the container is decreased.
Option c. the size of the container is decreased.Explanation:
At constant temperature, the pressure of a gas has the following relations with other variables:
Amount of gas: direct proportion, the greater the amount of particles of the gas, molecules or atoms, the greater the pressure of the gas.Then, as long as the number of molecules in the gas does not change, a decrease in the mass of the gas molecules (option a) does not modify the pressure, which makes that the option a. is not valid.
Option b, diffusion of the gas molecules increases, means that some molecules will abandon the container. So, following the direct proportion of the pressure with the number of molecules, this option means a decrease of the pressure, and you discard it.
Following the same reasoning, the option d, number of gas molceules in the container is decreased, also means a decrease of the pressure, and this option is discarded.
Volume: as per Boyle's law, the volume and the pressure of a gas are in inversed relation. Then, the option c., size of the container is decreased, indeed means the increase of the pressure, and this is the correct option.Question 2. When Gay-Lussac’s law of combining volumes holds, which of the following can be expressed in ratios of small whole numbers? a. pressures before and after reaction b. volumes of gaseous reactants and products c. kelvin temperatures d. molar masses of products and molar masses of reactants
option b. volumes of gaseous reactants and productsExplanation:
Gay-Lussac’s law of combning volumes states that, at constant temperature and pressure, when gases combine the volumes are in the ratio of simple whole numbers.
Hence, it is not pressures before and after reaction (option a), kelvin temperatures (option c), or molar masses of products and molar masses of reactants (option d) what holds.
It is volumes of gaseous reactants and products (option b) what holds.
Question 3. Equal volumes of ideal gases at the same temperature and pressure contain equal numbers of a. protons. b. ions. c. particles. d. electrons.
option c. particles.Explanation:
This is a direct deduction from Avogadro's principle: no matter the size of the particles, either single atoms, or small or large molecules, at the same temperature and pressure, equal volume of gases contain the same number of particles (atoms or molecules).
That is why it is stated that at 1 atm and 0°C, the volume of 1 mole of any gas is approximately 22.4 liter.
Question 4. At constant temperature and pressure, the volume of a gas is directly proportional to its a. molar mass. b. number of moles. c. density at STP. d. rate of diffusion.
option b. number of moles.Explanation:
As explained on the answer to the question 4, Avogadro's law states that at constant temperature and pressure, the volume of a gas is directly proportional to its number of particles.
Moles is a unit of amount of particles. One mole is equal to 6.022 × 10²³ particles (atoms or moles, in the case of gases).
You can also reason from the ideal gas equation:
pV = nRT ⇒ n = (pV) / (RT) = V (p / RT)Then, since (p / RT) is constant, p is directly proportional to V.