ameliaxbowen7
12.12.2019 •
Chemistry
The molar mass of table salt (naci) is 58.5 g/mol.
what mass of salt is equivalent to 3.00 moles naci?
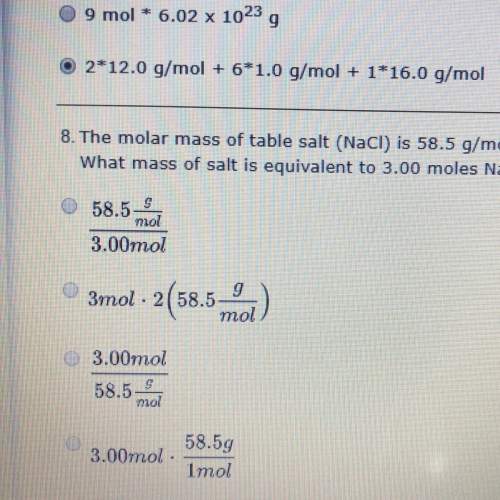
Solved
Show answers
More tips
- F Family and Home What is a Smart Home System and How Does it Work?...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- C Computers and Internet Log-in: what is it and why do you need it?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...

Ответ:
What is the atomic mass of hafnium if, out of every 100 atoms, 5 have a mass of 176, 19 have a mass of 177, 27 have mass of 178, 14 have a mass of 179, and 35 have a mass of a 180?
Chemistry Atomic Mass
Average atomic mass of Hafnium
=
178.55
amu
Explanation:
If there are two isotopes an element
Average atomic mass
=
mass
a
×
fract
a
+
mass
b
×
fract
b
In the given problem about Hafnium, 5 isotopes have been given.
Extending the above expression to 5 isotopes
We obtain
Average atomic mass
=
176
×
0.05
+
177
×
0.19
+
178
×
0.27
+
179
×
0.14
+
180
×
0.35
=
178.55
-.-.-.-.-.-.-.-.-.-.-.-.-.-
(Ensure that sum of all fractions adds to 1.
It is easy to find the fraction of each isotope in the question as total number is given as 100 atoms. Number of each isotope divided by 100)
Always use average atomic masses for mass calculations involving elements or compounds.