Foxfire5109
02.04.2021 •
Chemistry
The pyruvic acid from glycolysis can be converted to acetic acid, which can enter the citric acid cycle. Because this process is a cycle, the atoms entering the cycle must balance the atoms that leave. Consider carbon balance in the citric acid cycle. How many CO2 molecules leave the cycle for each acetic acid molecule that enters? For this question, assume that carbon only enters as acetic acid and exits as CO2 . Note that acetic acid has two carbon atoms. You can determine the number of carbon atoms in carbon dioxide from its formula, CO2 .
Solved
Show answers
More tips
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry Solution of lithium chloride and lead(ii) acetate are mixed stoichiometrically (a) write a balanced chemical equation for this reaction with the correct phases of all substances...
- C Chemistry Asurface condenser consists of 200 thin walled circular tubes (each tube is 22.5 mm in diameter and 5 m long) arranged in parallel, through which water flows. if the mass flow...
- C Chemistry Sulfuric acid is easily neutralized by potassium hydroxide to form water and a soluble salt. awrite a balanced chemical equation for this reaction with the correct phases of all...
- C Chemistry Design an ro wtp to meet the following criteria. influent feed tds- 3000 mg/l. finished water produced flow rate-1.5 mgd. tds in treated water- 300 mg/l. temperature of the feed-200....
- C Chemistry 123 005w of heat is removed from water vapour (at 373k) condensing on the outer surfaces (at 363k) of an array of cylinders of length 0.6m and diameter 0.05m. how many cylinders...
- C Chemistry Write a python program to parse the ncbi genbank search result, i.e., data 1eb file in the directory as example. use regular expression to parse the file and output into fasta...
- C Chemistry A6.55 g sample of aniline (c6h5nh2, molar mass = 93.13 g/mol) was combusted in a bomb calorimeter with a heat capacity of 14.25 kj/°c. if the initial temperature was 32.9°c, use...
- C Chemistry How do you improve the ductility of steel? explain how ductility is important for some of the manufacturing processes- name the processes...
- C Chemistry We have observed during the semester various reactions that are driven by the hydrolysis of ppi (pyrophosphate) by pyrophosphatase. ppi-h2o à 2 pi find and diagram out three reactions...
- C Chemistry An ion is an atom with a net electrical charge due to_?...

Ответ:
The correct answer is - 2CO2 or two molecules of carbon dioxide.
Explanation:
The TCA cycle or citric acid cycle is a central pathway of cellular respiration that helps in unifying several metabolites and generates energy for cellular functions. It takes place over eight different steps:
Step 1: Acetyl CoA enters the citric acid cycle that is a 2-carbon compound and attaches with 4-carbon compound oxaloacetate to form citrate a 6 carbon compound
Step 2: This 6-carbon compound isocitrate
Step 3: 5 carbon compound alpha-ketoglutarate is produced by oxidizing the isocitrate and release the release of carbon dioxide. One NADH molecule is formed.
Step 4: This 5 carbon molecule is oxidized again to form a four-carbon compound that binds with CoA to make forming succinyl CoA. A second molecule of NADH is produced, alongside a second molecule of carbon dioxide.
Step 5: Succinyl CoA is then converted to succinate with GTP formation
Step 6: Succinate is converted into fumarate with the generation of FADH₂ molecule.
Step 7: Fumarate is converted to malate
Step 8: oxaloacetate is formed with the NADH molecule.
Ответ:
The ratio of mass of sulfur atom to mass of oxygen atom in sulfur dioxide is 1: 1.
Explanation:
Law of constant proportion states that In a chemical substance the elements are always present in definite proportions by mass. This law is also known as 'Law of definite proportions '.
Mass of 1 atom of sulphur = 32 g
Mass of 1 atom of oxygen = 16 g
Mass of 2 atoms of oxygen =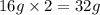
In formation of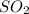 , 1 atom of sulfur combines with 2 atoms of oxygen and thus the mass ratio will be 32: 32= 1:1 .
, 1 atom of sulfur combines with 2 atoms of oxygen and thus the mass ratio will be 32: 32= 1:1 .
Thus the ratio of mass of sulfur atom to mass of oxygen atom in sulfur dioxide is 1: 1.