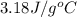The stability of atomic nuclei is related to the ratio of protons to electrons ratio of neutrons to protons number of protons and neutrons number of electrons
Solved
Show answers
More tips
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry BROOO PLEASE HELP ME...
- C Chemistry Now make the same calculations for a 1.0×10−11 m solution of hclo4. [h+] = ph = poh = [oh-] =...
- C Chemistry How can Redox reactions happen in covalently bonded compounds? A. Redox might happen but it’s not likely. B. Redox cannot happen because electrons are not being transferred. C. Redox...
- M Mathematics Monique got a loan where she repays $300 every month. The interest rate is 3.2%/a, compounded monthly. It will take her 6 years to pay off the loan. Which formula relates to this...
- M Mathematics The sum of three conservative integers is -204. what is the largest integer?...
- M Mathematics In ∆abc, ad is the angle bisector of ∠a and be is the angle bisector of ∠b. if ad intersects be at point o, find the measure of ∠aob if: c m∠a +m∠b = 110°...
- M Mathematics On two investments totaling $14,000, Peter lost 4% on one and earned 6% on the other. If his net annual receipts were $312, how much was each investment?...

Ответ:
The stability of atomic nuclei is related to the .
ratio of neutrons to protons . number of protons and neutrons.Ответ:
Answer : The specific heat of the metal is,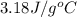
Explanation :
Heat released by the reaction = Heat absorbed by the calorimeter + Heat absorbed by the water
where,
q = heat released by the reaction
c = specific heat of metal = ?
m = mass of metal = 11.6 g
Now put all the given values in the above formula, we get:
Thus, the specific heat of the metal is,