cookiemonster0476
07.02.2021 •
Chemistry
The standard heat of combustion is shown in the following chemical equation CgH 20 (g) + 140 2(g) 9CO 2(g) + 10H 2 o (1) delta
Solved
Show answers
More tips
- H Health and Medicine What to Take with You to the Maternity Hospital?...
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- C Chemistry The phase change in which a substance changes from a solid to a liquid is a. freezing. b. melting. c. sublimation. d. condensation....
- C Chemistry What is the enthalpy of the overall chemical equation...
- C Chemistry Which is considered a cation? a. h+ b. cl- c. ne d. he...
- C Chemistry If the heat involved in a chemical reaction has a negative sign...
- C Chemistry Which type of weather technology often carries cameras that can make images of earth s surface clouds and snow cover?...
- C Chemistry The road trip you and your friend are planning is 1430km on way. What is the cost of the gasoline in US dollars for the round trip if fuel price is 1.403 euros per liter? 0.91 euro=1.00...
- C Chemistry A student titrates a solution containing 0.050 moles of calcium hydroxide with phosphoric acid. How many moles of phosphoric acid need to be added to reach the equivalence point?...
- C Chemistry Qual a classificação quanto ao número de hidróxido ?de um exemplo...
- C Chemistry 56ude67fv67gbubvtyfg help PLS...
- C Chemistry An equation is said to be balanced it follows the Law of Conservation of Mass a. true b. false...

Ответ:
The given question is incomplete. The complete question is:
The standard heat of combustion is shown in the following chemical equation
 . If 130 g of nonane combusts , how much heat is released?
. If 130 g of nonane combusts , how much heat is released?
6211.21 kJ
Explanation:
Heat of combustion is the amount of heat released on complete combustion of 1 mole of substance.
Given :
Amount of heat released on combustion of 1 mole of nonane = 6125.21 kJ
According to avogadro's law, 1 mole of every substance occupies 22.4 L at NTP, weighs equal to the molecular mass and contains avogadro's number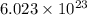 of particles.
of particles.
1 mole of nonane weighs = 128.2 g
weighs = 128.2 g
Thus we can say:
128.2 g of nonane on combustion releases = 6125.21 kJ
Thus 130 g of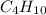 on combustion releases =
on combustion releases =
Thus the heat of combustion of 130 g of nonane is 6211.21 kJ
Ответ:
532 4934 2932r29023 94394392
Explanation:
i had that question