tfaulk2884
06.08.2019 •
Chemistry
The titrate 20.00 milliliters of an unknown hc2h3o2 smaple, use 43.00 of a standard 0.1 m naoh titrant. what is the molarity of the hc2h3o2?
0.215 m
0.1000m
860.0m
0.04651 m
Solved
Show answers
More tips
- F Family and Home Do You Really Need a Playpen for Your Child?...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- C Computers and Internet Log-in: what is it and why do you need it?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
Answers on questions: Chemistry
- C Chemistry ANSWER AND BE DE OF THE BUNCH In the chemical equation wA + xB --- yC + zD, if one knows the mass of A and the molar masses of A, B, C, and D, one can determine...
- C Chemistry PLEASE HELP I WILL GIVE YOU BRAINLIEST (the linked photo)!!! thank you...
- C Chemistry What is the symbol and number of protons in Arsenci...
- C Chemistry How many elements are there in the periodic...
- C Chemistry A solution is made by adding 23.1 mL of concentrated nitric acid ( 70.4 wt% , density 1.42 g/mL ) to some water in a volumetric flask, and then adding water to...
- S Social Studies True story: when I was 17, I was socially awkward at a party, so I stood in the kitchen next to one of those fridges with a built in water dispenser and just kept...
- P Physics Para rescatar a un niño de 22.9 kg en un incendio, un bombero se ajusta una bolsa de herramientas junto con su casco y sale por una ventana que se encuentra en...
- B Business Difference between partial equilibrium and general equilibrium in the simplest form...
- M Mathematics Select the equivalent expression. \left(5^{4}\cdot b^{-10}\right)^{-6}=?(5 4 ⋅b −10 ) −6 =?left parenthesis, 5, start superscript, 4, end superscript, dot, b, start...
- S Social Studies Which of the following best describes an ethical framework?...

Ответ:
0.1000M
Explanation:
Given parameters:
Volume of acid = 20mL, to liter gives 2 x 10⁻²L
Volume of NaOH = 43mL to liter gives 4.3 x 10⁻²L
Molarity of NaOH = 0.1M
Unknown:
Molarity of acid = ?
Solution;
The acid is easily identified as a carboxylic acid which is called ethanoic acid. With the identification of the acid, we can write the reaction equation as shown below:
CH₃COOH + NaOH → CH₃COONa + H₂
Now that we have established the reaction equation, we proceed to solve for molarity of the acid given.
To solve for the molarity of the acid, we solve from the known parameters to the unknown ones. We know the details of the base NaOH used.
We first find the number of moles of NaOH as shown below:
Number of moles of NaOH = molarity x volume of NaOH
Number of moles of NaOH = 0.1 x 4.3 x 10⁻² = 0.0043mole
From the reaction equation, we know that:
1 mole of NaOH reacted with 1 mole of ethanoic acid
0.0043mole of NaOH will produce 0.0043mole of the acid
Now to find the molarity of the acid, we use the expression below:
Molarity of acid =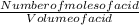
Molarity of acid =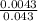 = 0.1M
= 0.1M
Ответ: