elwinelwin9475
18.08.2019 •
Chemistry
The value of δg°\' for the conversion of dihydroxyacetone phosphate to glyceraldehyde-3-phosphate (gap) is 7.53 kj/mol. if the concentration of dihydroxyacetone phosphate at equilibrium is 1.85 mm, what is the concentration of glyceraldehyde-3-phosphate? assume a temperature of 25.0 °c. the constant r = 8.3145 j/(mol·k)
Solved
Show answers
More tips
- G Goods and services What Are the Most Popular Services?...
- O Other What is the oldest joke ever told?...
- L Legal consultation How to Properly Inherit: Tips and Recommendations...
- C Computers and Internet Boost your processor performance with these easy tips...
- S Sport How does Bodyflex work: what is it and how does it work?...
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- S Style and Beauty How to Properly Apply Eye Makeup: Tips from a Professional Makeup Artist...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
- A Auto and Moto What is the Average Lifespan of an Engine in a Car?...
Answers on questions: Chemistry
- C Chemistry The strongest intermolecular forces of attraction exist in a liquid whose heat of vaporization is...
- C Chemistry If there are 6 moles of hydrogen and 2 moles of oxygen that are combined to make water, how many moles of water can be made?...
- C Chemistry Which two come together to form an ionic bond? a. a positive cation and a ngetaive cation b. a positive cation and a negative anion c. a positive cation and a positive anion...
- C Chemistry Which phase of matter does not have a definite shape yet the particles will not fill the container?...
- C Chemistry Para preparar el almuerzo en una cocina de gas se necesitan 12000 kJ, ¿cuánto dinero nos cuesta preparar una comida en una cocina de butano? ¿Y en una cocina de propano? Si tuviésemos...
- C Chemistry How many molecules are in 5.40 mol of CO2?...
- C Chemistry The image below shows the moon and the earth. at what time would moonrise happenbr /> around 9:00 AM around 3:00 PM around 9:00 PM around 3:00 AM...
- C Chemistry Write a balanced equation for the reaction between hydrogen peroxide H202 and Fe2+ to produce Fe3+ and H2O in acidic solution...
- C Chemistry Which electromagnet is stronger and why?...
- C Chemistry WILL MARK BRAINLIEST AND 30 POINTS A radioactive nuclide that is used to label blood platelets has 49 protons and 62 neutrons. Which is the symbol of this nuclide? Superscript...

Ответ:
The concentration of glyceraldehyde-3-phosphate is 0.0886 nM.
Explanation:
The chemical equation for the conversion follows:
The expression for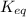 of above equation is:
of above equation is:
Let the concentration of glyceraldehyde -3-phosphate be 'x'
Relation between standard Gibbs free energy and equilibrium constant follows:
where,
R = Gas constant =
T = temperature =![25^oC=[273+25]K=298K](/tpl/images/0175/5173/0e82f.png)
[Glyceraldehyde-3-phosphate] = x
[Dihydroxyacetone phosphate] = 1.85 mM
Putting values in above equation, we get:
Hence, the concentration of glyceraldehyde-3-phosphate is 0.0886 nM.
Ответ:
sxdcghnkjkn
Explanation: