misswonderless
12.08.2020 •
Chemistry
There are eight consitutional isomers with the molecular formula C4H11N. name and draw a structural formulas for each amine.
Solved
Show answers
More tips
- C Cities and Countries Which Country has the Most Expensive Visa?...
- G Goods and services Don t Let Your Fridge Smell Bother You: How to Get Rid of Unpleasant Odors in Your Refrigerator...
- F Family and Home What does a newborn need?...
- F Food and Cooking How to Make Cottage Cheese Casserole? A Proven Recipe...
- H Health and Medicine How to Improve Eyesight: Science-based Techniques to Enhance Your Visual Acuity...
- A Auto and Moto What to expect from the new VW Golf 7?...
- F Food and Cooking Learn How to Make Ice Cream at Home - Step by Step Guide...
- F Food and Cooking Discover the most delicious spaghetti with these tips...
- F Food and Cooking Why Chicken Liver Pops and How It Can Affect Your Health?...
- F Food and Cooking How to Choose the Right Olive Oil: A Comprehensive Guide...

Ответ:
See figure 1
Explanation:
We have to remember that in the isomer structures we have to change the structure but we have to maintain the same formula, in this case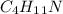 .
.
In the formula, we have 1 nitrogen atom. Therefore we will have as a main functional group the amine group.
In the amines, we have different types of amines. Depending on the number of carbons bonded to the "N" atom. In the primary amines, we have only 1 C-H. In the secondary amines, we have two C-N bonds and in the tertiary amines, we have three C-N bonds.
With this in mind, we can have:
-) Primary amines:
1) n-butyl amine
2) sec-butyl amine including 2 optical isomers
3) isobutyl amine
4) tert-butyl amine
-) Secondary amines:
5) N-methyl n-propyl amine
6) N-methyl isopropyl amine
7) N, N-diethyl amine
-) Tertiary amines:
8) N-ethyl N, N-dimethyl amine
See figure 1
I hope it helps!
Ответ:
I think not 100% sure, I think its D uppermost mantle.