23ricorvan
18.12.2019 •
Chemistry
This set contains the spectra of benzil, biphenyl, and bibenzyl, three compounds used in the melting point experiment. using your knowledge of ir spectroscopy and the structures of these three compounds, determine which spectrum belongs to which compound and give a brief (one sentence) explanation of your choice.
Solved
Show answers
More tips
- S Society and Politics Why Did Medvedev Announce a New Privatization of State Property?...
- F Family and Home Stay Warm but Don t Overheat: What is the Optimal Temperature for Your Home During Winter?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
Answers on questions: Chemistry
- C Chemistry Which of the following reactions have more entropy in the products than the reactants? a. c3h8(g) + 3o2(g) co2(g) + 4h2o(l) b. 2h2o(l) 2h2(g) + o2(g) c. 4ag(s) + o2(g)...
- C Chemistry How to separate mixture of small copper and iron beads?...
- C Chemistry A118-ml flask is evacuated and found to have a mass of 97.129 g. when the flask is filled with 768 torr of helium gas at 35 ? c, it is found to have a mass of 97.171...
- C Chemistry A series of logical steps that is followed in order to solve a problem is called the * 1 point A. experimental process. B. scientific theory. D. scientific method...
- C Chemistry Magnesium metal reacts with solid boron oxide (B203) to form elemental boron and magnesium oxide, which are both solids. What is the coefficient of boron in the balanced...
- C Chemistry Calculate the number of moles there are in 3.6 x 10^23 molecules of AlBr3...
- C Chemistry The mass percentage composition of cryolite, a compound used in the production of aluminum, is 32.79% Na, 13.02% Al, and 54.19% F. What is the empirical formula of...
- C Chemistry What causes an element from the periodic table to be more or less reactive?...
- C Chemistry An isotope of platinum has 100 neutrons. What is the atomic number of this specific isotope of platinum? 100, 98, 195, 198...
- C Chemistry A mixture is made by combining 1.61 lb of salt and 5.37 lb of water. What is the percentage of salt (by mass) in this mixture?...

Ответ:
Spectrum 1- Biphenyl
Spectrum 2 - Benzil
Spectrum 3 - Bibenzyl
Explanation:
For Benzil:
The Spectrum - 2 shows this compound.The C=O and C-H bond stretching observed at following values.
For Biphenyl:
Spectrum - 1 shows this compound.
There are stretching vibrations from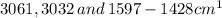 due to C-H stretching and C-C stretching respectively of aromatic ring only.
due to C-H stretching and C-C stretching respectively of aromatic ring only.
For Bibenzyl:
Spectrum - 3 shows this compound
There are stretching vibrations at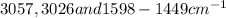 due to C-H stretching and C-C stretching of aromatic ring.
due to C-H stretching and C-C stretching of aromatic ring.
Along with this, stretching vibrations at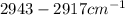 due to C-H of alkyl.
due to C-H of alkyl.
Ответ:
A: 0.67
The situation described in the question is analogous to a semipermeable membrane. Water is able to pass through aquaporin channels present in the liposome, but large uncharged particles (glucose) and ions (K+ and Cl -) are impermeable and will remain trapped within the liposome. If assumed to be ideal, the osmotic pressure, π, exerted by the solution due to molarity differences across the membrane is defined as π = iMRT, where i is the van't Hoff factor, M is the molarity of the solution, R is the universal gas constant, and T is the absolute temperature of the solution. A change in osmotic pressure at constant temperature is due to changes in iM, a term that is equivalent to the concentration of dissolved particles produced by solute in solution. When compared to the original volume of 1 M glucose, the new combined solution has twice the volume and three times the number of dissolved particles (1 M KCl, a strongly electrolytic solution, produces 1 M concentrations of both K+ and Cl- in solution), or an increase in the concentration of dissolved particles by a factor of 1.5. This is equivalent to a combined molarity of dissolved particles of 1.5 M. The ratio of osmotic pressure is then [1 M dissolved glucose] / [1.5 M dissolved glucose + KCl] = 0.67
Explanation: