Two atoms With diferent mass number but the same atomic number are called
Solved
Show answers
More tips
- G Goods and services How to Choose a Humidifier? Helpful Tips and Recommendations...
- W Work and Career What is the Most In-Demand Profession in the Modern World?...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- F Food and Cooking Red Caviar: How to Choose the Best?...
- C Computers and Internet Clearing Cache: How to Speed Up Your Browser...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- C Chemistry The following results were obtained during the Gravimetric and Volumetric determination of iron in sample A. Gravimetric: 29.18, 29.10, 29.08, 29.17, and 29.04. Volumetric:...
- C Chemistry Write four facts of cross breeding plants...
- C Chemistry Lead can be prepared from galena (lead II sulfide) by roasting the galena in the presence of oxygen to form lead II oxide and sulfur dioxide. Heating the metal oxide with more...
- C Chemistry Help help help help help identify the tupe of bond...
- C Chemistry 19.9% of all the isotopes of an element have a mass of 9.93 amu and 80.1% have a mass of 10.94 amu. What is the average mass of this element?...
- C Chemistry In observance of Dalton’s Law given the Patm = 102.63kPa and the PH2O= 1.4kPa T=?...
- C Chemistry Hi, can i have help please? Calculate the pOH, [H+1], and the [OH-] of rainwater pH = 6.2...
- C Chemistry Why were the transformed cells plated on media containing kanamycin and not ampicillin?...
- C Chemistry How were the ideas of Aristotle and Democritus similar to the alchemists ?...
- C Chemistry 4- Imagine que, em seu município, exista um relógio de sol (gnomon) muda,dependendo da hora do dia claro e da Época do ano, responda. a- como ela varia ao longo de um dia?em...

Ответ:
☃️ Chemical formulae ➝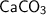
How to find?
For solving this question, We need to know how to find moles of solution or any substance if a certain weight is given.
And, Then we need to find the number of molecules which can be found by:
Here, Na = Avagadro Number (6.022 × 10²³)
☀️ As it is a compund, We considered molecules. Then, we can predict the no. of atoms of elements in one molecule of compound and get the answer.
Solution:
Atomic weight of elements:
Ca = 40C = 12O = 16❍ Molecular weight of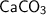
= 40 + 12 + 3 × 16
= 52 + 48
= 100 g/mol
❍ Given weight: 10 g
Then, no. of moles,
⇛ No. of moles = 10 g / 100 g mol‐¹
⇛ No. of moles = 0.1 moles
☄ No. of moles of Calcium carbonate in the given weight = 0.1 moles
Finding no. of molecules,
⇛ No. of molecules of CaCO3 = 0.1 × 6.022 × 10²³
⇛ No. of molecules of CaCO3 = 6.022 × 10²²
Now,
1 molecule ofThen, 6.022 × 10²² molecules will have,
= 3 × 6.022 × 10²² atoms of oxygen
= 18.022 × 10²² atoms of oxygen
In more scientific form,
= 1.8022 × 10²³ atoms of oxygen
☄ Hence, Final answer - 1.8022 × 10²³ atoms
━━━━━━━━━━━━━━━━━━━━