mzlennon639
05.06.2020 •
Chemistry
Un elemento posee dos isótopos cuyos números de masa suman 68 y la semisuma de sus neutrones es 19. Determine el grupo de este elemento en la Tabla Periódica Actual.
Solved
Show answers
More tips
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- S Style and Beauty How to Get Rid of Peeling Nails: Natural Remedies...
- S Science and Technology Understanding Magnetic Storms: A Guide to This Natural Phenomenon...
- F Family and Home What is Most Important in Men s Lives?...
- G Goods and services Which TV is better - LCD or Plasma?...
- C Computers and Internet Are there special gaming mice?...
- G Goods and services LED-подсветка в LCD-телевизорах: 5 причин, почему она лучше других технологий...
- C Computers and Internet Keep Your Mouse Pad Clean: The Right Way to Clean It?...
- C Computers and Internet 3D Glasses! What is this thing?...
Answers on questions: Chemistry
- C Chemistry Question 16 (1 point) Find the number of grams in 29.32 mol ammonia (NH3). Round your answer to two decimal places and be sure to include the proper units. Your Answer Answer...
- C Chemistry Here for no reason. ahhhhhhhhhhhhhhhhhhhhhh...
- C Chemistry 640 C of electricity is passed through an electrolytic cell containing a solution of zinc ions. Calculate the amount (in mol) of zinc that will be formed at the cathode. (Faraday...
- C Chemistry Describe how electromagnetic waves are formed and travel through space....
- C Chemistry Elements are simple ___ made of the same type of . They cannot be ___ down chemically. There are about 118 known elements and most elements are ___ They are in appearance and...
- C Chemistry What is the exact atomic mass of 53X...
- C Chemistry What mass (grams) of carbon disulfide will react with 5.3 x 10 molecules of oxygen?...
- C Chemistry 7. Which letter indicates the most gradual change in temperature? 8. Which letter indicates the most sudden change in temperature? 9. Why are the average temperatures in the middle...
- C Chemistry Hey mamas rateme 1-10 ;)))))jdjdbjdjsjsjsbsb...
- C Chemistry What is permafrost? a. dry desert soil c. ice on the tundra b. muddy soil d. soil that is always frozen...

Ответ:
The element belongs to group 15 of the present periodic table
Explanation:
Isotopes have the same proton number, what differs is that they have different number of neutrons.
Now since the half-sum of their neutrons is 19, the total number of neutrons would be 19 * 2 = 38 neutrons.
Also, their masses add up to be 68, since the total mass equals sum of neutrons and sum of protons, this means that the total number of protons is 68-38 = 30
Since this is equal in both isotopes, this means that the proton number of the element is 30/2 = 15
The atomic number is the number of protons in the nucleus of an atom. Since the atomic number is 15, this element is phosphorus and it belongs to group 15 of the present periodic table
Ответ:
The compound in which the atom has unfilled valence shell of electron is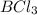
Explanation:
Valence electrons is defined as the electrons which are present in the outermost shell of an atom.
Lewis-dot structure is defined as the structure which represents the number of valence electrons around the atom. The dots represents the electrons.
For the given options:
Option 1:In the given compound, 2 electrons are shared between oxygen and each hydrogen atom. From the lewis dot structure,
Valence electrons in each hydrogen atom = 2
Valence electrons in oxygen atom = 8
The valence shell of both the elements is complete.
Option 2:In the given compound, 4 electrons are shared between carbon and each oxygen atom. From the lewis dot structure,
Valence electrons in each oxygen atom = 8
Valence electrons in carbon atom = 8
The valence shell of both the elements is complete.
Option 3:In the given compound, 2 electrons are shared between carbon and each hydrogen atom. From the lewis dot structure,
Valence electrons in each hydrogen atom = 2
Valence electrons in carbon atom = 8
The valence shell of both the elements is complete.
Option 4:In the given compound, 2 electrons are shared between boron and each chlorine atom. From the lewis dot structure,
Valence electrons in each chlorine atom = 8
Valence electrons in boron atom = 6
The valence shell of boron is not complete and has incomplete octet.
Hence, the compound in which the atom has unfilled valence shell of electron is