drejones338p04p2p
10.07.2019 •
Chemistry
Use enthalpies of formation given in appendix c to calculate δh for the reaction br2(g)→2br(g), and use this value to estimate the bond enthalpy d(br−br).
Solved
Show answers
More tips
- C Computers and Internet How to Top Up Your Skype Account Without Losing Money?...
- H Health and Medicine How does childbirth happen: everything future mothers need to know...
- H Health and Medicine What Are the Best Vitamins? A Scientific View on Vitamin Supplements...
- A Auto and Moto How to Deregister a Car in Moscow?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- A Auto and Moto How to choose the right drive for your BMW...
- H Health and Medicine 10 Tips for Avoiding Vitamin Deficiency...
- L Leisure and Entertainment Mysteries of the Name Vyacheslav Dobrynin...
- H Health and Medicine What makes Graves’ disease dangerous?...
- F Food and Cooking When is Easter in 2011?...
Answers on questions: Chemistry
- C Chemistry At a certain temperature the rate of this reaction is first order in HI with a rate constant of 0.0632 s 2HI g = H2 g + I2 g Suppose a vessel contains HI at a concentration of 1.28M....
- C Chemistry 1. Identify the seven diatomic elements and explain why they are different from the rest of the elements...
- C Chemistry Garfield (weighing 24 lbs) took a flight to the moon on the space shuttle. as usual, he stuffed himself with lasagna during the entire flight and napped when he wasn t eating. much...
- C Chemistry Liquid water at 200 kpa and 15°c is heated in a chamber by mixing it with superheated steam at 200 kpa and 150°c. liquid water enters the mixing chamber at a rate of 4.3 kg/s, and...
- C Chemistry Is the formation of sulfur tetrafluoride monoxide endothermic or exothermic?...
- C Chemistry Which organism belongs to eubacteria?...
- C Chemistry if an object has a density of 2.5 g/cm3 and it is placed in a tank of water, will the object float or sink in water...
- C Chemistry Is 2H2O(l) - 2H2(g) + O2(g) exothermic or endothermic?...
- C Chemistry PLEASE HELP 50 POINTS Instructions: Follow the directions below to complete part one and part two of this activity. You will submit both parts once completed. Note: You are not performing...
- C Chemistry Match the label to the correct description. ball-and-stick model advantage ball-and-stick model disadvantage space-filling model advantage space-filling model disadvantage...

Ответ:
Given reaction represents dissociation of bromine gas to form bromine atoms
Br2(g) ↔ 2Br(g)
The enthalpy of the above reaction is given as:
ΔH = ∑n(products)Δ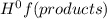 - ∑n(reactants)Δ
- ∑n(reactants)Δ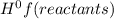
where n = number of moles
Δ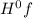 = enthalpy of formation
= enthalpy of formation
ΔH = [2*ΔH(Br(g)) - ΔH(Br2(g))] = 2*111.9 - 30.9 = 192.9 kJ/mol
Thus, enthalpy of dissociation is the bond energy of Br-Br = 192.9 kJ/mol
Ответ:
So the formula of circumference is (diameter x Pi) or (2r x Pi).
In this case, your diameter is 2 so your equation would be 2 x 3.14= 6.28 and rounded to the nearest tenth would your answer would be 6.30 inches.
Step-by-step explanation: