ggdvj9gggsc
13.07.2019 •
Chemistry
Using the following equation 5kno2+2kmno4+3h2so4=5kno3+2mnso4+k2so4+3h20. starting with 2.47 grams of kno2 and excess kmno4 how many grams of water will be produced?
Solved
Show answers
More tips
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Science and Technology Colliders: How They Work and Why They Matter...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
Answers on questions: Chemistry
- C Chemistry I m having trouble understanding what the difference in energy between the products and reactants from the surroundings means. Can you help explain what this means? please...
- C Chemistry 30. Phản ứng nào dưới đây không phải là phản ứng thế? A. CuO + H2 → Cu + H2O B. Mg +2HCl → MgCl2 +H2 C. Ca(OH)2 + CO2 → CaCO3 +H2OD. Zn + CuSO4 → ZnSO4 +Cu 31. Trong số các chất...
- C Chemistry Describe what happens to the particles when a substance: 1. Melts 2. Freezes 3. Boils 4. Condenses...
- C Chemistry Who prepared aqua regia ?...
- C Chemistry Cho 0.62 g P tác dụng vừa đủ với m g dung dịch H2SO4 98%.Sau phản ứng hoàn toàn thu được V lít khí SO2 (đktc là sp khử duy nhất) viết pthh tính m,V...
- C Chemistry En els sòlids, les forces de cohesió són molt predominen sobre les de repul- sió. Les partícules del cos es- tant a prop entre elles i es mantenen en fixes. De totes maneres,...
- C Chemistry LiOH + KCl → LiCl + KOH The reaction above should theoretically yield 35.5 grams of LiCl. When Tom actually completes the reaction in the lab, he produced only 6 grams of LiCl....
- C Chemistry Which of the following is an example of how a scientist might use a model? a) a stopwatch is used to time the rate of a chemical reaction b) the end of a slinky is moved vertically...
- C Chemistry Which of these elements has four total electrons in its outer p orbitals? barium (ba) nitrogen (n) silicon (si) sulfur (s)...
- C Chemistry In this lab we will build an instrument called a coffee cup calorimeter: identify the list with all the necessary parts...

Ответ:
Given equation :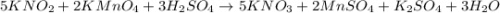
Given information = 2.47 grams KNO2 and excess KMnO4 and we need to find grams of water (H2O).
Since KMnO4 is in excess, so grams of water(H2O) can be calculated using grams of KNO2 with the help of stoichiometry.
To find grams of water(H2O) from grams of KNO2 , we need to follow three steps.
Step 1. Convert 2.47 grams of KNO2 to moles of KNO2.
Molar mass of KNO2 = 85.10 g/mol
Moles = 0.0290 mol KNO2
Step 2. Convert moles of KNO2 to moles of H2O using mole ratio.
Mole ratio are the coefficient present in front of the compound in the balanced equation.
Mole ratio of KNO2 : H2O is 5 : 3 (5 coefficient of KNO2 and 3 coefficient of H2O)
Mole = 0.0174 mol H2O
Step 3. Convert mole of H2O to grams of H2O
Grams = Moles X molar mass
Molar mass of H2O = 18.00 g/mol
Grams of water = 0.313 grams H2O
Summary : The above three steps can also be done in a singe setup as shown below.
In the above setup similar units get cancelled out and we will get grams of H2O as 0.313 grams water (H2O)
Ответ:
4 weeks change = -20
1 week change = -5
If x is the temperature at the start of the week:
A: (4x-20)/4
B: Change per week = x-5 This means that the temperature goes down by 5 degrees per week, with x being the temperature at the start of the week.
Read more on -