Ackussinglake63
16.06.2020 •
Chemistry
Using the solubility of Ca(IO3)2 that you determined in 0.01M KIO3 as a solvent, calculate the value of Ksp using only concentrations, that is assuming that all activity coefficients are 1.0. You should report 3 significant figures.
Solved
Show answers
More tips
- H Health and Medicine What are Electronic Cigarettes? The Ultimate Guide...
- C Computers and Internet How to Get Rid of 3pic Infector: Everything You Need to Know...
- A Auto and Moto Experience the World of the Most Expensive Cars on the Planet...
- S Style and Beauty How to Get Rid of a Double Chin?...
- F Food and Cooking How to Cook Julienne? Recipes and Tips...
- D Dating, Love, Relationships 10 Useful Tips on How to Survive a Breakup?...
- F Food and Cooking Apple Cider Vinegar: The Ultimate Health and Beauty Solution...
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- C Chemistry Mitchell is creating a model of a plant cell. Mitchell wants to include organelles that are involved in energy production for the cell. Which organelles should Mitchell include?...
- C Chemistry 3.5 Quiz: Chemical Reactions Please help me...
- C Chemistry Idon t understand this 2a+3b— c if 4.0 mol of a is reacted with 4.0 mol of b which of the reactant is limiting a) b is limiting because 4. 0 mol and 6. 0 mol are needed. b) a...
- C Chemistry Will be the brainiest guaranteed. emily wants to show that she knows the shapes of galaxies. she takes a tennis ball and makes a small cut in it to release the air inside. she...
- C Chemistry How do scientists know how much of a product can be formed?...
- C Chemistry what is the mass in grams of a block of gold measuring 6.4 cm x 12.5 cm x 26 cm if the density of gold is 19.3 g/cm3?...
- C Chemistry What is the measurement of the average molecular kinetic energy of a substance....
- M Mathematics Is 23in bigger than 2ft...
- M Mathematics What does 2w=1370 mean...
- M Mathematics One half X +13 equals nine...

Ответ:
Explanation:
Dissociation of Ca(IO₃)₂ is as follows:
Ca(IO₃)₂ ⇄ Ca²⁺ + 2IO₃⁻
This implies that for every mole of Ca(IO₃)₂ that dissolves in water' 1 mole of Ca²⁺ is obtained and 2 moles of IO₃⁻ is obtained.
The solubility in mol/L when Ca(IO₃)₂ is added to KIO₃ = 0.00341
Concentration of Ca²⁺ will be = 0.00341 M ˣ 1 = 0.00341 M ( i.e equal to the solubility of Ca(IO₃)₂)
Concentration of IO₃⁻ will be = 0.00341 M ˣ 2 = 0.00682 M
IO₃⁻ is also obtained from the dissociation of KIO₃ in water. the IO₃⁻ obtained from this process is = 0.01 M
The total concentration of IO₃⁻ now = 0.00682 M + 0.01 M = 0.01682 M
The solubility product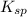 can be calculated by the formula:
can be calculated by the formula:
Ответ:
A compressed spring
Explanation: