Barbiealice8362
03.07.2019 •
Chemistry
We know that for a given reaction when the temperature increases from 100 k to 200 k the rate constant doubles. what is the activation energy in kj/mol
Solved
Show answers
More tips
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
Answers on questions: Chemistry
- C Chemistry Astudent did an experiment with two identical plants, plant 1 and plant 2. about 10 ml fertilizer was added to both plants. about 200 ml of water was added to plant 1...
- C Chemistry 2mno−4(aq)+5so2(g)+2h2o(l)→2mn2+(aq)+5so2−4(aq)+4h+(aq) what are the reduction and oxidizing agents...
- C Chemistry If you have hydrogen and oxygen what can you make?...
- C Chemistry What are the uses of petroleum?...
- C Chemistry Concentrated HCl is 98% and has density 1.46g/cm 3 what volume of concentrated acid required to make 2L of 0.25M HCl ....
- C Chemistry Astatin tidak digunakan di dalam makmal . jelaskan...
- C Chemistry Por qué el punto de ebullición de los alcoholes varia menos que el punto de fusión?...
- C Chemistry Which of the following is the correct model of C6H14?...
- M Mathematics Hay esta ayudame plissss y te doy corona si me ayudas....
- S SAT Which of the following is not an enumerated power of what congress can do:...

Ответ:
The activation energy of the reaction is 1.152 kJ/mol.
Explanation:
Activation energy is the minimum amount which is absorbed by the reactant molecules to undergo chemical reaction.
Initial temperature of reaction =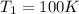
Final temperature of reaction =
Initial rate of the reaction at 100 k =
Final rate of the reaction at 200 k =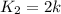
Activation energy is calculated from the formula:
R = Universal gas constant = 8.314 J/ K mol
Ответ: