mpete1234567890
16.04.2020 •
Chemistry
What a paragraph about what is calorimetry and how it works. Describe an experiment where you use a calorimeter to determine the heat capacity of a substance.
Solved
Show answers
More tips
- H Health and Medicine How to Deal with Heat Stroke?...
- F Food and Cooking Discover the Health Benefits of Cedar Nuts...
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- S Science and Technology Colliders: How They Work and Why They Matter...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport Playing Bowling: Rules and Advice for Novices...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
Answers on questions: Chemistry
- M Mathematics Please help quickly its super late...
- M Mathematics Find the exact value sin 79° cos 19° - cos 79° sin 19°...
- A Advanced Placement (AP) Calcula. La. Diagonales de un cuadrado que tiene de altura 6 cm...
- B Biology How can messages from the endocrine system reach cells throughout the body...

Ответ:
Calorimetry is the science or act of measuring changes in state variables of a body for the purpose of deriving the heat transfer associated with changes of its state. One could easily calculate the heat capacity of a substance by creating an experiment. This can be done by first gathering deionized water to measure the heat exchange between the metals and styrofoam cups which will serve as the calorimeter. Put these on a weigh scale and make sure to zero it. Afterward, add 100 mL of deionized water to the cups, find the mass of the water afterword accurately. Next, get 50 mg of copper metal and put it in a test tube within the beaker and measure it on the scale accurately. Next, heat the metal, but do not allow the beaker to get below the level of the metal in the test tube. Once the water is boiling, transfer it to the calorimeter. Be sure to remove the drop of water on test tube with towel, but do this quickly to avoid cooling. Next, transfer the metal to the calorimeter and cover. One should swirl the solution to be sure a full heat exchange happened. Be sure to dry the metal quickly before putting it in the calorimeter. Furthermore, measure the solution temperature to the first decimal place. This will complete the experiment and one should easily be able to calculate the heat capacity of the substance. Overall, their are many experiments that can measure a substance's heat capacity, but this experiment specifically measures the heat capacity of various metals using a simple calorimeter.
Explanation:
Be sure to check for grammatical errors, but overal idea should be good.
Ответ:
a)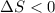
b) entropy of the sistem equal to a), entropy of the universe grater than a).
Explanation:
a) The change of entropy for a reversible process:
The energy balance:
If the process is isothermical the U doesn't change:
The work:
If it is an ideal gas:
Solving:
Replacing:
Given that it's a compression: V2<V1 and ln(V2/V1)<0. So:
b) The entropy change of the sistem will be equal to the calculated in a), but the change of entropy of the universe will be 0 in a) (reversible process) and in b) has to be positive given that it is an irreversible process.