Bladedrose2351
18.05.2021 •
Chemistry
What are the acids and bases? Thanks!
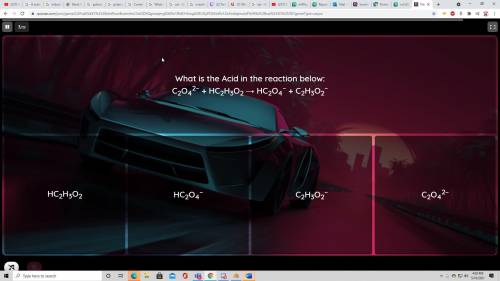
Solved
Show answers
More tips
- C Computers and Internet What Are Peers and Seeds in Torrenting?...
- H Health and Medicine 10 Simple Techniques on How to Boost Your Mood...
- G Goods and services How to Choose the Right High Chair for Your Baby?...
- S Style and Beauty Learn how to tie a keffiyeh on your head like a pro...
- S Style and Beauty How to braid friendship bracelets?...
Answers on questions: Chemistry
- C Chemistry N2+h2- nh3. so you know you need to balance this out....
- C Chemistry Solve? commission %-5 sale -$23.325.00...
- C Chemistry The temperature of a hot air balloon is increased. What would happen to the density of the balloon?...
- C Chemistry How many ppm of C29H60 are in 23 pm C29H60? ( 9.4 ppm)Can anyone explain it?...
- C Chemistry When an atom forms negaitive ion. Give the number of valence, 70. Using orbital notation, 75. Which has the greater lattice energy, 81. Give the formula for each,...
- C Chemistry Disadvantage of nitrogen cycle...
- C Chemistry Hace 18 mil millones de años, el Ylem (A) estalló, expandiéndose y formándose las moléculas de hidrógeno y helio (B), que dieron origen después a las estrellas...
- C Chemistry Formula for trichlorine tetroxide...
- C Chemistry Balance the equation below_AgO— _Ag+_O2...
- H History What do these headlines say about how the Supreme Court has addressed the principal of equality on which the United States was founded?...

Ответ:
an acid is a molecule or ion capable of donating a proton or alternatively capable of forming a covalent bond with an electron pair
bases are substance which read with acid as originally proposed by G.F Rouelle in the mid-18th century
AND YOUR WELCOME
Ответ:
D. 77.5.
Explanation:
From the balanced equation: CaCl₂ + 2AgNO₃ → 2AgCl + Ca(NO₃)₂It is clear that 1.0 mole of CaCl₂ reacts with 2.0 moles of AgNO₃ to produce 2.0 moles of AgCl and 1.0 mole of Ca(NO₃)₂.The no. of moles of reacted (30.0 g) CaCl₂ = mass / molar mass = (30.0 g) / (110.98 g/mol) = 0.27 mol.Using cross multiplication:
1.0 mole of CaCl₂ produces → 2.0 moles of AgCl, from the stichiometry.
0.27 mol of CaCl₂ produces → ??? moles of AgCl.
∴ The no. of moles of the produced AgCl = (2.0 mol)(0.27 mol) / (1.0 mol) = 0.54 mol.
∴ The mass of the produced AgCl = no. of moles x molar mass = (0.54 mol)(143.32 g/mol) = 77.48 g ≅ 77.5 g.