allimaycatp8qgaq
23.12.2019 •
Chemistry
What is δg′° for the reaction ( k ′ eq measured at 25 °c)? (b) if the concentration of fructose 6-phosphate is adjusted to 1.5 m and that of glucose 6-phosphate is adjusted to 0.50 m, what is δg? (c) why are δg′° and δg different?
Solved
Show answers
More tips
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- C Computers and Internet How to Set Up Internet on iPhone? Detailed Guide with Step-by-Step Instructions...
- P Philosophy 8 привычек, чтобы достичь счастливой жизни...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- H Health and Medicine How to Choose the Right Glasses?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...

Ответ:
a.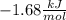
b.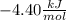
c. Standard conditions provide 1 M molarity for each component, while non-standard conditions provide specific concentrations
Explanation:
a. The original problem states that:
For an equilibrium:
This means:
Solving for the standard Gibbs free energy change:
b. Now we will solve for the non-standard Gibbs free energy change given by the equation:
Here:
We obtain:
c. The standard Gibbs free energy change is measured for 1 M concentration of each molarity, however, the non-standard Gibbs free energy change is measured for the given molarities of 1.5 M and 0.50 M. Due to different conditions, we obtain different values.
Ответ: