What is the atmoic number for potassium?
Solved
Show answers
More tips
- H Health and Medicine How to Deal with Heat Stroke?...
- F Food and Cooking Discover the Health Benefits of Cedar Nuts...
- S Science and Technology Exploring Our Galaxy: How Many Planets are in the Milky Way?...
- S Science and Technology Colliders: How They Work and Why They Matter...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment How Many Seasons are There in the TV Show Interns?...
- S Sport Playing Bowling: Rules and Advice for Novices...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- S Sport How to Learn Swimming? Simple Tips for Beginners...
- C Computers and Internet What is Web 2.0 and How Does it Work?...
Answers on questions: Chemistry
- S Social Studies 3) Read through the timeline. Number the following events in chronological order, one being the farthest back in history: Arab Spring began. Suez Canal built United States withdrew...
- C Chemistry This occurs because of slower moving particles due to decreased energy A. boiling B. condensation C. melting D. all of these...
- E English Dad Jokes 101: Why are frogs so happy? They eat whatever bugs them. (Bu-Dum-Tss)...
- B Biology Which of the following is a calm area on Earth s surface where warm air rises and there is little or no wind? Question 2 options: Land Breeze Doldrums Polar Easterlies Trade Winds...

Ответ:
19
Explanation:
Ответ:
0.668 g of barium sulfate
Explanation:
Given,
Balanced chemical equation: BaCl₂ + Na₂SO₄ → BaSO₄ + 2NaCl.
Volume of BaCl₂ = 25.34 mL x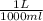 = 0.02534 L.
= 0.02534 L.
Molarity of BaCl₂ = 0.113 M
Molarilty =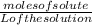
Moles of solute = Molarilty x L of the solution
Moles of BaCl₂ = 0.113 M x 0.02534 L = 0.00286 mol.
From the balanced chemical equation there is a 1:1 molar ratio between BaCl₂ and BaSO₄
Therefore, moles of BaCl₂ = moles of BaSO₄
Moles of BaSO₄ = 0.00286 mol.
Mass of BaSO₄ = moles of BaSO₄ x Molar mass of BaSO₄
Mass of BaSO₄ = 0.00286 mol x 233.4 g/mol.
Mass of BaSO₄ = 0.668 g.