joselynrobles
04.07.2019 •
Chemistry
What is the average speed (actually the root-mean-square speed) of a argon atom at 27°c?
Solved
Show answers
More tips
- S Science and Technology Colliders: How They Work and Why They Matter...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- M Mathematics Cheryl writes 7 fewer posts than Kevin on a social media site. Answer the questions to write an algebraic expression. Then evaluate the expression for the given value. Which operation...
- B Business A restaurant owner in the 1950s does not feel negatively toward African Americans but refuses to serve them in his restaurant because he fears that doing so would offend customers....
- C Chemistry The decomposition of 11.0 g of fe2o3 results in...

Ответ:
The average speed (actually the root-mean-square speed) of the argon atom is 432.8 m/s
The thermodynamic temperature T is proportional to the average kinetic energy of a molecule. The mean translational kinetic energy of an atom or (molecule) of an ideal gas is proportional to the thermodynamic temperature.
That is, the average speed (actually the root-mean-square speed)
1/2m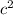 = 3KT/2
= 3KT/2
Where
m = molar mass
c = speed
K = Bolzman constant
T = Temperature.
Given that the temperature of a argon atom is 27°C
Mass of an argon m = 6.63 x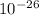 Kg
Kg
Bolzman constant K = 1.38 x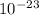 J/K
J/K
Temperature T = 273 + 27 = 300K
Substitute all the parameters into the equation
1/2m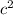 = 3KT/2
= 3KT/2
m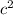 = 3KT
= 3KT
c =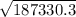
c = 432.8 m/s
Therefore, the average speed (actually the root-mean-square speed) of the argon atom is 432.8 m/s
Learn more here: link
Ответ:
Option (D) is the correct answer.
Explanation:
When we use an object or substance in excess then it is likely that the substance will run out with time. But at the same time, if we use that substance according to its requirement and try to conserve it as much as we can then we can preserve the substance.
Hence, it is good to avoid wastage of a substance and balance its use according to the need.
Therefore, we can conclude that in the given situation yes, there is a problem with the solution as the need to conserve copper must be balanced with use.