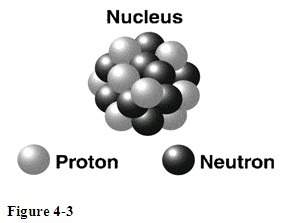
What is the change on the subatomic particles represented in figure 4-3? assuming all the particles in the nucleus are visible, what are the atomic and mass numbers of the atom shown?
Solved
Show answers
More tips
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- G Goods and services What Are the Most Popular Services?...
- O Other What is the oldest joke ever told?...
- L Legal consultation How to Properly Inherit: Tips and Recommendations...
- C Computers and Internet Boost your processor performance with these easy tips...
- S Sport How does Bodyflex work: what is it and how does it work?...
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- S Style and Beauty How to Properly Apply Eye Makeup: Tips from a Professional Makeup Artist...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
Answers on questions: Chemistry
- C Chemistry Plesseee help me pretty pleaee...
- C Chemistry Crossing a Dd parent with another Dd parent results in...
- C Chemistry The cells of all organisms must produce energy in order for the cell to survive and function. The diagrams below show different parts of an animal cell and a plant cell....
- C Chemistry What is the strength of alloys and what is the solubility of alloys? please please DO NOT SEARCH I ALREADY SEARCHED AND THERE IS NO ANSWER THAT I AM LOOKING FOR!!...
- C Chemistry an ion m3+ contain 10 electron and 14 neutron. what are the atomic number and mass number of the element M...
- C Chemistry Which of the following states of matter has a definite volume...
- C Chemistry PLEASE HELP!! IM SO CONFUSED!...
- M Mathematics 10. Simplify, (1/2^2/3 x 3^-2)...
- M Mathematics A number divided by 7 is a minimum of 10...
- H History PLZ HELP SIMPE TRUE OR FALSE The tribes in Pontiac s Conspiracy came only from the south, near Georgia true or false The Stamp Act was the first tax to visibly affect...

Ответ:
Explanation:
Atomic number means the number of protons an atom holds. Whereas mass number equals (number of protons + number of neutrons).
The given figure shows that there are 9 neutrons and 8 protons.
Therefore,
mass number of the atom = number of protons + number of neutrons
= 8 + 9
= 17
Hence, mass number of the atom is 17 and atomic number of the atom is 8.
Ответ:
Explanation:
The given data is as follows.
Refractive index of mixture = 1.456
Refractive index of hexane = 1.375
Refractive index of toulene = 1.497
Let mole fraction of hexane =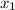
and, mole fraction of toulene =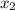
Also,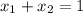
or,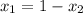
Hence, calculate the mole fraction of hexane as follows.
refractive index mixture= mole fraction hexane × ref index hexane + mole fraction toluene × ref index toluene.
1.456 =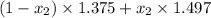
1.456 =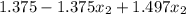
0.081 =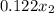
= 0.66
Since,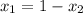
= 1 - 0.66
= 0.34
Thus, we can conclude that mole fraction of hexane in your sample is 0.34.