tjeffers90028
24.12.2019 •
Chemistry
What is the effect of adding more co2 to the following equilibrium reaction? co2 + h2o h2co3
a. more h2co3 is produced.
b. more h2o is produced.
c. the equilibrium is pushed in the direction of reactants.
d. no change
also,
which of the following is the correct expression for the rate of the following reaction? note: all species are gaseous.2so3 < --> 2so2 + o2a. [so3]2 / ([so2]2 x [o2])
b. [so2]2 x [o2] / [so3]
c. [so2]2 x [o2] / [so3]2
d. [so2] x [o2] / [so3]
Solved
Show answers
More tips
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- L Leisure and Entertainment What can be considered an antique: defining and valuing old objects...
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- C Computers and Internet Step-by-Step Guide on How to Download Music to Your iPhone...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- F Family and Home How to Choose Suitable Windows for Your Home?...
- L Leisure and Entertainment What Movies You Should Watch: A Guideline to Make the Right Decision...
- L Leisure and Entertainment How to Identify a Song? Which Program Can Recognize a Song by Its Melody?...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- C Chemistry Research the Greenhouse effect and how it relates to heat transfer....
- C Chemistry Plsss help will mark BRAINLIEST...
- C Chemistry Question 1 You are making lemonade and the calls for 6 cups Lemon juice (L), 3 Cups of sugar (S) and 5 cups of water (W) to make 12 Cups of Lemonade (LA): your formula is: 6 L +...
- C Chemistry Base your answers to questions 22, 23, and 24 on the information below and on your knowledge of chemistry. The diagrams represent four different atomic nuclei. 22. Identify the...
- C Chemistry Convert 1351 ps into ns...
- C Chemistry Which one of the following compounds is insoluble in water? a. BaSO4 b. KNO3 c. Na2SO4 d. Li2CO3...
- M Mathematics The goal of this exercise is to study euler’s important identity e ix = cos(x) + isin(x) and some of its consequences that will be useful in this course. (a) find the real and imaginary...
- B Business Pindyck & rubinfeld, 8e. ch. 11, #9. you are an executive for super computer, inc. (sc), which rents out super computers. sc receives a fixed rental payment per time period...
- B Business Today, thomas deposited $100,000 in a three-year, 12% cd that compounds quarterly. what is the maturity value of the cd? (a) $309,090.(b) $119,410.(c) $109,270.(d) $142,576....
- B Business Purchase date # of units cost per unit total cost 4/17 beginning inventory 2,000 $115 $230,000 6/1 beginning inventory 700 $120 $84,000 8/22 beginning inventory 1,000 $130 $130,000...

Ответ:
1) A. More H2CO3 is produced.
2) C.![K=\frac{[SO_2]^2\times[O_2]}{[SO_3]^2}](/tpl/images/0431/4779/57391.png)
Explanation:1) Any change in the equilibrium is studied on the basis of Le-Chatelier's principle.
This principle states that if there is any change in the variables of the reaction, the equilibrium will shift in the direction to minimize the effect.
If concentration of is increased, that is the reactant is increased, so according to the Le-Chatlier's principle, the equilibrium will shift in the direction where decrease of concentration of
is increased, that is the reactant is increased, so according to the Le-Chatlier's principle, the equilibrium will shift in the direction where decrease of concentration of  takes place. Therefore, the equilibrium will shift in the right direction, i.e.
takes place. Therefore, the equilibrium will shift in the right direction, i.e. 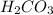 will be formed.
will be formed.
2) The rate of a equilibrium reaction is determined by equilibrium constant. Equilibrium constant is the ratio of the product of the concentration of products to the product of the concentration of reactants each term raised to their stochiometric coefficients.
Ответ:
The intermolecular bonding in the solid is van der Waals dispersion forces. That means that it will have a lower melting point than diamond or graphite where you have giant covalent bonding. In fact C60 sublimes at 800 K (527°C). Diamond and graphite sublime at around 4000 K.
Explanation: