jessec27019
14.05.2021 •
Chemistry
What is the final concentration of Ca(OH)2
when 255 mL of 0.250 M Ca(OH)2 is mixed
with 55.0 mL of 0.65 M Ca(OH)2?
Solved
Show answers
More tips
- G Goods and services Is an Air Ionizer Necessary for Your Home?...
- C Construction and repair How to Choose the Best Underfloor Heating?...
- C Computers and Internet How to Get Rid of Windows Genuine Check?...
- C Computers and Internet War of Social Media: Which Platform is the Leader?...
- H Health and Medicine How to Treat the Flu: A Comprehensive Guide...
- O Other What is a Disk Emulsifier and How Does it Work?...
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
Answers on questions: Chemistry
- C Chemistry Below are some of the steps completed in the titration process. (i) add base from the buret to the acid. (ii) transfer a known volume of acid to an erlenmeyer flask. (iii)...
- C Chemistry Briefly workout the relationship between these constants: In consideration of the decopmposition of hydrogen iodide....
- C Chemistry Modern periodic table is based upon atomic number. Explain what does the modern periodic table states about the atomic numbers ?...
- C Chemistry Consider the reaction of 0.113 g of aluminum metal with 50.0 mL of 0.235 M Co(NO₃)₂. How many moles of Co(NO₃)₂ do you start with?...
- C Chemistry Which correctly lists two characteristics of crystal faces that define crystal systems? angle and number number and color size and hardness size and angle...
- C Chemistry At 0 half-lives, a sample of Uranium-238 is 60 grams. After one half-life has gone by, how many grams of uranium-238 will remain? A.60 grams B.15 grams C.30 grams D.7.5...
- C Chemistry Convert 8.42 x 1024 atoms of aluminum into moles. (AKS 4e)...
- C Chemistry Edelman and Gronkowski want to play a game of beach volleyball. If the beach ball has a volume of 261 L at a temperature of 502 K, what will the temperature of the balloon...
- C Chemistry What percent of the world s population has flown on an airplane? 80% 60% 40% less than 10%...
- B Biology Which of the following statements is true? the mineral ores for gold and silver are commonly found in the earth s crust. the mineral ores for gold and silver are rarely...

Ответ:
20 drops of ethanol dispensed by dropper will be 1.0 mL ethanol.
Explanation:
Mass of 15 drops of ethanol = 0.60 g
Mass of 1 drop of an ethanol =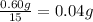
Mass of 1.0 mL of ethanol = m
Volume of ethanol = 1.0 mL
Density of an ethanol = d = 0.80 g/ml
Number of drops in 0.80 g of ethanol = x
20 drops of ethanol dispensed by dropper will be 1.0 mL ethanol.