What is the hybridization of the central atom in each of the following? 1. beryllium chloride 2. nitrogen dioxide 3. carbon tetrachloride 4. xenon tetrafluoride
Solved
Show answers
More tips
- D Dating, Love, Relationships Does a Person s Character Depend on the Color of Their Eyes?...
- O Other Childhood Fears: What Many of Us Experienced...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- L Leisure and Entertainment What to Bring on a Hike? Essential Items to Pack for a Safe and Enjoyable Adventure...
- L Leisure and Entertainment Couchsurfing: A New Way to Travel...
- S Style and Beauty Autotanning: Harmful or Safe?...
- F Food and Cooking 10 Ideas for a Wedding Anniversary Gift...
- H Health and Medicine How to Reduce Sweating in the Heat and Beyond: Say Goodbye to Excessive Sweat...
- F Food and Cooking Do Aphrodisiacs Really Work? Separating Fact from Fiction...
- H Health and Medicine What to Eat to Lose Weight?...
Answers on questions: Chemistry
- C Chemistry The nearest star is 4.2ly away from earth. how far is this in miles...
- C Chemistry Ahypothetical element has two main isotopes with mass numbers of 62 and 65. if 66.00% of the isotopes have a mass number of 62 amu, what atomic weight should be listed...
- C Chemistry Which variable determines the electron energy in the hydrogen atom according to bohr’s model...
- C Chemistry Calcium oxide (cao) can react with carbon dioxide (co2) in a synthesis reaction. select the most likely products of this reaction. cao + co2 → ? caco + o2 caco3 ca + co2...
- C Chemistry Which statement best compares coastal ecosystems to open ocean ecosystems? coastal ecosystems include abyssopelagic depths, and open ocean ecosystems do not. coastal ecosystems...
- C Chemistry What phrase describes a good scientific question? has a wide focus addresses a gap in knowledge does not lead to a testable hypothesis O has a simple yes or no answer vious...
- C Chemistry B) Clasifica las siguientes reacciones químicas apareando con el número que le corresponda. 1- Síntesis NaOH + HCL → NaCL + H2O 2-Descomposición AgNO3 + KCL → KNO3 +AgCL...
- C Chemistry Which of these is an example of a chemical change?...
- C Chemistry 2. a solution in which 50 grams of salute is dissolved in 250 grams of solution has a concentration of...
- C Chemistry Which of the following atoms has the highest electronegativity? use the periodic table to answer this question....

Ответ:
Answer :
(1) The hybridization of central atom beryllium in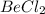 is,
is, 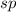
(2) The hybridization of central atom nitrogen in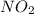 is,
is, 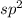
(3) The hybridization of central atom carbon in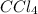 is,
is, 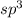
(4) The hybridization of central atom xenon in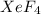 is,
is, 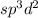
Explanation :
Formula used :
where,
V = number of valence electrons present in central atom
N = number of monovalent atoms bonded to central atom
C = charge of cation
A = charge of anion
Now we have to determine the hybridization of the following molecules.
(1) The given molecule is,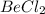
The number of electron pair are 2 that means the hybridization will be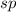 and the electronic geometry of the molecule will be linear.
and the electronic geometry of the molecule will be linear.
(2) The given molecule is,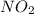
If the sum of the number of sigma bonds, lone pair of electrons and odd electrons present is equal to three then the hybridization will be,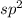 .
.
In nitrogen dioxide, there are two sigma bonds and one lone electron pair. So, the hybridization will be,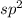 .
.
(3) The given molecule is,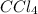
The number of electron pair are 4 that means the hybridization will be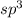 and the electronic geometry of the molecule will be tetrahedral.
and the electronic geometry of the molecule will be tetrahedral.
(4) The given molecule is,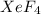
Bond pair electrons = 4
Lone pair electrons = 6 - 4 = 2
The number of electrons are 6 that means the hybridization will be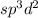 and the electronic geometry of the molecule will be octahedral.
and the electronic geometry of the molecule will be octahedral.
But as there are four atoms around the central xenon atom, the fifth and sixth position will be occupied by lone pair of electrons. The repulsion between lone and bond pair of electrons is more and hence the molecular geometry will be square planar.
Ответ:
vdhdjdbes
Explanation:
shve eueevev