allison9746
25.11.2020 •
Chemistry
What is the mass (in grams) of a CO_2 gas sample that occupies 5.46 L at STP?
Solved
Show answers
More tips
- F Food and Cooking How to Make the Perfect Glühwein: Step-by-Step Guide...
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- F Food and Cooking How many stages of coffee roasting are there?...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- F Food and Cooking Experts Name Top 5 Healthiest Teas...
- C Computers and Internet Google Search Tips and Tricks: Everything You Need to Know...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Liver Cleansing - Rejuvenation for the Body?...
- S Style and Beauty Is Photoepilation the Solution to Unwanted Hair Forever?...
Answers on questions: Chemistry
- C Chemistry Initially, you have two containers with 10 mL of water at 20°C. David decides to heat up the water, while Chris cools the water. Who would have a greater volume of water?...
- C Chemistry Alison wants to record a song she wrote. The music studio records the music using digital technology instead of analog technology. Why does the music studio likely use digital...
- C Chemistry A given sample of metal has a density of 2.5 g/cm3 and a mass of 12.5 g. What is the volume of the metal? 31.25 cm^3 5.0 cm^3 0.20 cm^3 5.0 cm...
- C Chemistry Determinar los mililitros de solución 0.5 Normal de NaCl que se pueden preparar a partir de 3 L de solución de cloruro de sodio 1.5 Normal...
- C Chemistry The pH of 0.259 M solution of an unknown acid is measured to be 2.353. What is the Ka of the acid?...
- C Chemistry I need help with these if yall could please helpp a girl out cause I took this test 1 time nd boii- when i tell you I failed that I mean by I got a 0% on thatt Imaoooo but...
- C Chemistry 3. Which is the minimum amount of energy required to start a chemical reaction? A endothermic energy B chemical energy C activation energy OD exothermic energy...
- C Chemistry Which of the following has the highest vapor pressure? A) Ethanol at 25 Degrees Celsius B) Ethanol at 40 Degrees Celsius C) Ethanol at 50 Degrees Celsius D) Ethanol at 75...
- C Chemistry Write the balanced COMPLETE ionic equation for the reaction when Na₂CO₃ and AgNO₃ are mixed in aqueous solution. If no reaction occurs, write only NR....
- C Chemistry What is the molarity of a solution that contains 1.78 moles of KNO3 dissolved in 2.50L of water?...

Ответ:
10.7g of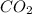
Explanation:
Any gas at STP occupies 22.4 L/mol.
As such:
5.46/22.4 = 0.244 mol of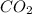
mass = mol * molecular weight = 0.244 * 44 = 10.7g of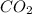
Ответ:
n2h4 + 7 h2o2 --> 2hno3 + 8h2o
so assuming that n2h4 is in excess and only moles of h2o2 are determining the product content then 7 moles of h2o2 give 2 moles of hno3
so:
7: 2
x: 0.08
x=0.28 moles of h2o2