What is the mass of 3.0 x 10^23 atoms of neon
Solved
Show answers
More tips
- B Business and Finance How to Open an Online Store? A Detailed Guide for Beginners...
- L Leisure and Entertainment What can be considered an antique: defining and valuing old objects...
- L Leisure and Entertainment Choosing the Right Books to Read: Tips and Recommendations...
- C Computers and Internet Step-by-Step Guide on How to Download Music to Your iPhone...
- H Health and Medicine Naskol ko Opasen Ukus Kleshcha i Kak Ego Raspoznat...
- F Family and Home How to Choose Suitable Windows for Your Home?...
- L Leisure and Entertainment What Movies You Should Watch: A Guideline to Make the Right Decision...
- L Leisure and Entertainment How to Identify a Song? Which Program Can Recognize a Song by Its Melody?...
- H Health and Medicine Coughing: Causes, Types, and Treatment Methods...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- S Social Studies How did the following groups view the Haitian Revolution? • American slave owners • Black slaves in America • French Revolutionaries • Absolute monarchs in Europe...
- M Mathematics Iwill love you forever if you do...
- C Chemistry Someone help please! Extra points & brainlest!...
- E English Read the following moral tale: patty the milkmaid was going to the market carrying her milk in a pail on her head. as she walked along, she began calculating what she would...

Ответ:
The mass of neon for the given number of atoms will be 9.8 g.
Explanation:
We are given:
Number of neon atoms =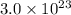
According to the mole concept:
So,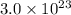 will be occupied in =
will be occupied in = 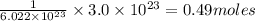
To calculate the number of moles, we use the equation:
Moles of Neon = 0.49 moles
Molar mass of neon = 20 g/mol
Putting values in above equation, we get:
Hence, the mass of neon for the given number of atoms will be 0.49g.
Ответ:
Explanation:
tdhghsrxdjtfkgyhgykuftr