Officialavm
26.05.2021 •
Chemistry
What is the mass of NH4Cl that must dissolve in 100. grams of water at 50.°C to make a saturated solution?
Solved
Show answers
More tips
- F Family and Home What does a newborn need?...
- F Family and Home Choosing the Right Car Seat for Your Child: Tips and Recommendations...
- F Food and Cooking How to Get Reconfirmation of Registration?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
Answers on questions: Chemistry
- M Mathematics Justify the steps taken to justify the system...
- M Mathematics Which equation best represents the relationship between x and y in the graphic...
- M Mathematics 3. Statement: If three points are on the same line, then they are collinear. Points F, U, N are on line d. Conclusion: 4. Statement: If an animal is a frog, then it is an amphibian....
- E English Tomatoes from the garden make the best salsa.which best identifies the adjective phrase and the word it modifies in the sentence? a- tomatoes; modifies gardenb- from the garden;...
- H Health What is nutrition analysis of a recipe? a. information on mypyramid food groups included in the recipe b. information on the recipe s nutrient and calorie content c. information...

Ответ:
The oxidation half reaction for the given reaction is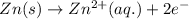
Explanation:
Oxidation reaction is defined as the reaction in which a substance looses its electron. The oxidation state of the substance gets increased.
Reduction reaction is defined as the reaction in which a substance gains electrons. The oxidation state of the substance gets reduced.
For the given chemical equation:
Oxidation half reaction: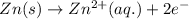
Reduction half reaction: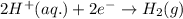
Zinc is getting oxidized and hydrogen is getting reduced.
Hence, the oxidation half reaction for the given reaction is