toricarter00
25.05.2020 •
Chemistry
What is the molarity of a NaOH solution if 15.0 mL is exactly neutralized
by 7.5 mL of a 0.02 M HC2H3O2 solution?
Show all work using titrations formula.
Solved
Show answers
More tips
- H Health and Medicine What to Take with You to the Maternity Hospital?...
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
Answers on questions: Chemistry
- H History Which argument did many americans make against the alien and sedition acts? a. they violated the constitutional guarantee of a free press. b. they were unfair to native-born...
- B Biology Predict what would happen to the H+ ion concentration if you added sulfuric acid to water. Would the pH increase or decrease...
- B Biology Choose the law that best explains the example: a wagon accelerating more slowly once mass is increased while force remains constant is an example of newton’s group of...
- B Biology Characteristics of the plasma membrane include which of the following? cellulose phospholipids and protein increased strength to the cell selective memebrane...
- M Mathematics IMPORTANT HW What is the translation from f(x) = 1x + 5 to g(x) = 3x + 5? A. Moved down B. Moved up C. Clockwise D. Counterclockwise...

Ответ:
see below for explanation
Explanation:
1. you need to set up and balance the equation
NaOH + HC2H3O2 --> NaC2H3O2 + H2O
2. Write the value they gave you underneath the equation to figure out what we are solving for, which is the molarity of NaOH
NaOH (Base) + HC2H3O2 (Acid)--> NaC2H3O2 + H2O
15.0 ml | 7.5ml
? M | 0.02M
3. Use the MaVa=MbVb ·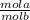
- This formula is saying the M of an acid times the volume of the acid = the M of the base times the volume of the base times the mol of the acid/ mol of the base ratio. ***You should always compute the volume in Liters.
- Convert your values (x is the molarity of the base, the unknown value) Our acid values are first because that is how the equation is set up
0.0075 · 0.02 = 0.0150 · x · 1/1
4. Simplify
0.00015 = 0.0150x
5. Solve
Molarity of the base = 0.01
Ответ:
Hey mate
after solving the inequality you found option A is correct
thanks