amandasantiago2001
02.10.2020 •
Chemistry
What is the most reasonable inference you could make about sulfur (S), which has four stable isotopes and an average atomic mass of 32.06 u?
Solved
Show answers
More tips
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- G Goods and services What Are the Most Popular Services?...
- O Other What is the oldest joke ever told?...
- L Legal consultation How to Properly Inherit: Tips and Recommendations...
- C Computers and Internet Boost your processor performance with these easy tips...
- S Sport How does Bodyflex work: what is it and how does it work?...
- H Health and Medicine How to Whiten Teeth and Get the Perfect Smile...
- S Style and Beauty How to Properly Apply Eye Makeup: Tips from a Professional Makeup Artist...
- A Auto and Moto How Can Parking Sensors Help Drivers?...
- C Computers and Internet Make Money Online: Secrets and Essential Ways...
Answers on questions: Chemistry
- C Chemistry What happens to the atoms in a scoop of ice cream as it melts?...
- C Chemistry Which element is most metallic in character? explain your reasoning...
- C Chemistry Silver nitrate reacts with aluminum chloride to form the insoluble compound, silver chloride. The reaction proceeds according to the balanced equation below: Wayne reacted 1.565...
- C Chemistry Compare the calculated percent ionization of acetic acid in the solution containing both 0.10 M acetic acid and 0.10 M sodium acetate to the calculated percent ionization for the...
- C Chemistry What would happen if a TLC plate was placed in a jar where the solvent level was above the level of the origin (sample spot)? What should you do if you inadvertently spotted your...
- C Chemistry H2so3 spell out the full name of the compound....
- C Chemistry With a manual transmission, the speed of the vehicle determines...
- C Chemistry The temperature at which water condenses from a gaseous to a liquid form...
- M Mathematics 10. If m10 = 77º, m7 = 47° and m 16 = 139, find each angle measure. a. m1 = f. m6= k. m13= b. m2= g. m8= I. m14= c. m3= h. m9= m. m15 = d. m4= i. m11= n. m17= e. m5= j. m12= o....
- E English What do you mean by haunted???do ghosts or paranormal beings exist in real??...

Ответ:
The most reasonable inference you could make about sulfur ( S ) is ; The most common isotope of sulfur is S-32
Although your question lacks some data a general answer is provided
Sulphur is a non-metallic chemical element with an atomic number of 16 and an average atomic mass of 32.06 u
The four stable isotopes of sulfur and their proportion are as follows :
S-32 ≈ 95%
S-33 ≈ 0.75%
S-34 ≈ 4.21%
S-35 ≈ 0.02%
Therefore we can infer that based on composition the most common isotope of sulfur is S-32.
Hence we can conclude that from the various isotopes compositions the most common isotope of sulfur is S-32.
Learn more : link
Ответ:
Sulphur is found in 23 isotopes ranging from 27 to 49.
Also, magnitude of average atomic mass is more toward the isotope which is the most abundant.
Now , molecular mass of Sulphur is 32.06 u . It means the most abundant isotopes of Sulphur is 32.
It can also be verified by the fact that 95 % of natural occurring sulfur is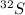 .
.
Hence, this is the required solution.
Ответ:
convalent bonds are stronger than ionic bond
Explanation:
it's a sharing of bond