frandariusscott
26.11.2019 •
Chemistry
What is the name of this hydrocarbon?
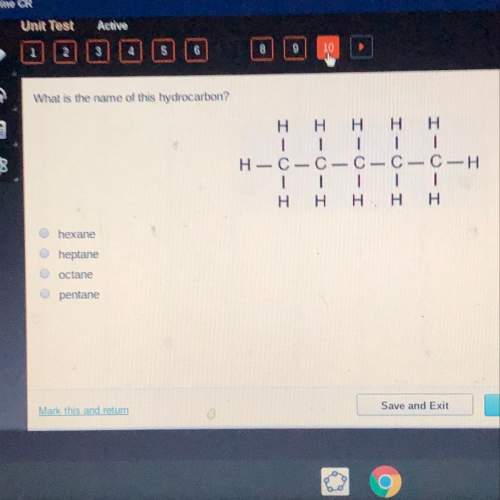
Solved
Show answers
More tips
- F Family and Home When and how to start introducing solid foods to your baby?...
- B Business and Finance Moneybookers – What it is and How it Works...
- C Computers and Internet How to Format Your C Drive: Detailed Guide and Tips...
- F Food and Cooking What can and cannot be eaten during Lent?...
- H Health and Medicine What to Do When Your Jaw Locks Up?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- D Dating, Love, Relationships Is it a Compliment or Flattery: What s the Difference?...
- S Science and Technology The Metric System in Our Daily Life: Understanding Its Importance...
- C Computers and Internet What to Do If Your ICQ Gets Hacked?...
- C Computers and Internet How to Choose a Monitor?...
Answers on questions: Chemistry
- C Chemistry Mg + 2HCI - MgCl2 + H2 If 500g of magnesium chloride (MgCl2) is produced in the above reaction, how many grams of hydrogen (H2) would be produced?...
- C Chemistry I really need answers please helpp...
- C Chemistry How many grams of magnesium are in sample containing 9.12+ 10 ^23atoms...
- C Chemistry Mg + 2HCI → MgCl2 + H2 How many grams of hydrochloric acid (HCI) is needed to produce 200g of H2?...
- C Chemistry A sample of 0.3207 g of an ionic compound containing the bromide ion (Br) is dissolved in water and treated with an excess of AgNO3. If the mass of the AgBr precipitate...
- C Chemistry At STP, 111.0 mL of a certain gas has a mass of 0.156 g. What is a possible identity of this gas? Select one:o Cl2O O3o C₂H6o O₂o N₂...
- C Chemistry Plz help me solve this question is it A,B,C or D...
- C Chemistry Jrkvffjhbyyjhbjnnjbnjbthjnbhbu...
- C Chemistry 1. When 0.990g of Na3PO4.12H20 reacts with excess BaCl2.2H20 how manymoles of Ba3(PO4)2are produced?...
- C Chemistry PA + 502 → P4010 If 400g of P4010 is needed for an experiment, how much P4 would you have to burn?...

Ответ:
answer:
hexance
explanation:
Ответ:
answer:
the answer is 4th point, pentane
explanation:
hexane - c6h14
heptane - c7h16
octane - c8h18
pentane - c5h12
Ответ:
1. reflected energy and melting sea ice*
2. The institutions gathered enough data for scientists to draw a conclusion about whether the annual global temperature is gradually increasing.
The data gathered by each institution is very similar to the data gathered by the other institutions.
Explanation:
The reflection of extra heat causes the sea ice to melt. This causes the sea ice to melt. This becomes a problem because the polar ice melts and contributes to the increase in the water content in the sea. This causes the rise in the sea levels.
The options (1) and (2) are correct because the institutions gathered enough data to reach plausible conclusions.