rileyeddins1010
05.01.2020 •
Chemistry
What is the percent composition by mass of nitrogen in the compound n2h4 (gram-formula mass = 32 g/mol)?
Solved
Show answers
More tips
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- L Leisure and Entertainment Carving: History and Techniques for Creating Vegetable and Fruit Decorations...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine Reasons for the Appearance of Warts: Everything You Need to Know...
- A Art and Culture How to Learn Screaming: Step-by-Step Guide for Beginners...
- H Health and Medicine Contraceptive Pills After 35: The Importance Of Choosing The Right Medication...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- G Geography Who wants to friend me...
- E English Custody determines whom the child will live with. a. legal b. joint or shared c. physical d. sole...
- C Chemistry Any form of water that falls to the earth from the sky. a.precipitation b.humidity c.wind d.none of the above...
- B Biology The average number of bones in the adult skeleton is...

Ответ:
Explanation:
1. Calculate the molar mass of N₂H₄
2N = 2 × 14 = 28
4H = 2 × 1 = 4
Tot. = 32
2. Calculate the mass percent of N
Ответ:
The percentage composition of the nitrogen in N₂H₄ is 87.5%
The percentage composition of an element in a compound can be obtained by using the following formula:
Percentage =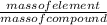 × 100
× 100
With the above formula, we can obtain the percentage composition by mass of nitrogen in N₂H₄ as illustrated below:
Molar mass of N₂H₄ = 32 g/mol
Mass of N in N₂H₄ = 2N = 2 × 14 = 28 g/mol
Percentage composition of Nitrogen =?Percentage =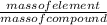 × 100
× 100
Percentage of nitrogen = × 100
× 100
Percentage of nitrogen = 0.875 × 100
Percentage of nitrogen = 87.5%Therefore, the percentage composition of the nitrogen in N₂H₄ is 87.5%
Learn more: link
Ответ: