Kelshonti15
09.02.2021 •
Chemistry
What is the precipitate in the following reaction?
AgNO3(aq) + KCl(aq)
AgCl(s) + KNO3(aq)
Solved
Show answers
More tips
- P Philosophy Agnosticism: Opinion or Belief?...
- C Computers and Internet How to Calibrate Your Monitor: Useful Tips and Recommendations...
- H Health and Medicine Hangover: How to Get Rid of It Quickly?...
- A Auto and Moto How to Start a Diesel Engine in Cold Weather?...
- A Auto and Moto Which alarm system to choose?...
- P Photography and Videography What is lens calibration and why is it needed?...
- S Science and Technology How to Choose a Picture Frame: Tips and Recommendations...
- P Photography and Videography Understanding HDR: How It Works and Why You Need It...
- P Photography and Videography How to Choose the Perfect Photo Paper for Your Images?...
- C Computers and Internet How to Choose an Uninterruptible Power Supply (UPS) for Your Computer: Expert Tips...
Answers on questions: Chemistry
- P Physics 3. Forces do always cause an object to move. need this ASAP...
- E English Re-read lines 392 - 407. What effect does the setting change have on the characters? Cite evidence to support your answer. To either side of the car, stretching into...
- M Mathematics Monique and her dad built a hang glider together. after testing the new glider for safety, the glider was ready to unfirst big night monique jumped off a tall sand...
- M Mathematics How old is your plz hurry and answer i need numbers...

Ответ:
The given information gives us the clue that deuterium and tritium react together. We need to find the balanced nuclear reaction of deuterium and tritium; however, first, let me explain you what deuterium and tritium are.
Deuterium and tritium are two isotopes of hydrogen having atomic mass of 2 and 3 respectively. Deuterium is represented by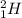 , whereas tritium is written as
, whereas tritium is written as 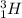 .
.
When deuterium and tritium collide together, they form Helium atom, which is written as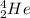 , and also release energy (and shoot out one neutron).
, and also release energy (and shoot out one neutron).
Hence, the balanced nuclear reaction will be the following:
-i