purplefive85
19.03.2020 •
Chemistry
What is the pressure in millimeters of mercury of 0.0130 molmol of helium gas with a volume of 210. mLmL at 55 ∘C∘C? (Hint: You must convert each quantity into the correct units (LL, atmatm, molmol, and KK) before substituting into the ideal gas law.)
Solved
Show answers
More tips
- S Society and Politics Is It Fact or Fiction? Let s Talk About Anton Chekhov s Pseudonym...
- S Sport Playing Bowling: Rules and Advice for Novices...
- C Computers and Internet How to Properly Repartition a Hard Drive?...
- A Auto and Moto What Is the Cost of Customs Clearance for a Car in Russia?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
Answers on questions: Chemistry
- B Biology MULTIPLE CHOICE QUESTION Which of these would not be an example of reproductive isolation? Male beetles evolve different shaped penises that no longer fit“ with the females reproductive...
- M Mathematics DA is tangent to the circle at A and DC is tangent to the circle at C. Find m...
- H Health What is the first step when making a decision? Select all that apply. A) use instincts and emotions B) list factors you want and compare C) identify the problem D) Do what your parents/guardians...

Ответ:
Answer : The pressure of the helium gas is, 1269.2 mmHg
Explanation :
To calculate the pressure of the gas we are using ideal gas equation:
where,
P = Pressure of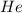 gas = ?
gas = ?
V = Volume of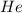 gas = 210. mL = 0.210 L (1 L = 1000 mL)
gas = 210. mL = 0.210 L (1 L = 1000 mL)
n = number of moles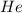 = 0.0130 mole
= 0.0130 mole
R = Gas constant =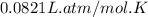
T = Temperature of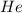 gas =
gas = 
Putting values in above equation, we get:
Conversion used : (1 atm = 760 mmHg)
Thus, the pressure of the helium gas is, 1269.2 mmHg
Ответ: