silveryflight
23.07.2020 •
Chemistry
WHAT IS THE UNBALCNED EQUATION OF Methane burns in oxygen to produce carbon dioxide and water
Solved
Show answers
More tips
- F Food and Cooking Which Calamari Salad is the Most Delicious?...
- S Style and Beauty How are artificial nails removed?...
- S Style and Beauty Secrets of Tying a Pareo: 5 Ways...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry 8. As you read, water continually moves from Earth s surface to the atmosphere through what processes?...
- C Chemistry How are you all take points...
- C Chemistry Isotonic compound have high melting point. Why ? Name the ions present in CaS. Pls answer fastly .. ...
- C Chemistry find the ratio of the initial volume to final volume if the temperature of the gas is changed from 27°C to 127°C at constant pressure....
- C Chemistry What effect would a decrease or increase in barometric pressure have on the boiling point...
- C Chemistry the volume of a gas at a pressure P is 100cm3. Assuming the temperature remains constant, find the new volume when the pressure is halved....
- C Chemistry Asalt bridge is used to provide electrons a path to flow from the electrode of one half-cell to another. true false...
- C Chemistry Which of the following uses a non renewable energy resource to generate energy ? a.) biomass power plant b.) wind farm c.) fireplace that burns wood d.) nuclear power...
- C Chemistry Ais a large molecule consisting of many repeating structural units. a. monomer c. polymer b. isomer d. elements...
- M Mathematics The number of cups of sugar (y) in a recipe increases by 2 for every increase of 4 cups of flour (x)....

Ответ:
Explanation:
Part 1: Naming compound formulas given the namesStep 1. Methane's formula is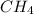 .
.
Step 2: Oxygen is a diatomic molecule (it exists bonded to itself for stability purposes), so by itself in chemical equations, it is written as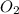 .
.
Step 3: Carbon dioxide is the molecular compound of one atom of carbon and two atoms of oxygen →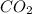 .
.
Step 4: Water is the common name of the compound of two hydrogen atoms and one oxygen atom →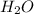 .
.
Part 2: Writing the skeleton equationStep 1: Use the determined formulas for the reactants and plug them into the equation. We are told that methane burns in oxygen -- hinting at a combustion reaction. Therefore, we may infer that these are the reactants that yield the products.
Skeleton equations are written with the reactant(s) on the left -- if there are several, they are separated by an addition symbol (+).
With this information, we may begin our equation: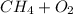 , where
, where 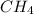 is methane and
is methane and 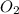 is the diatomic molecule of oxygen.
is the diatomic molecule of oxygen.
Step 2: Use the determined formulas for the products and plug them into the equation. We are told that the methane burns in oxygen to produce carbon dioxide and water. Hence, we can separate these two as we did with the reactants.
Now, our products side of the reaction will look like this: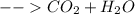 , where
, where 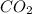 is carbon dioxide and
is carbon dioxide and 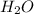 is water.
is water.
Step 3: Write the final equation. All you must do after determining both sides of the equation is simply push them together. Place the reactant side of the equation on the left and the product side of the equation on the right.
This gives us our final equation,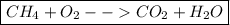 .
.
Because the problem asks for the unbalanced equation, we do not need to take any further steps of balancing the equation.
Ответ:
Annie gives the key to helen
helen lifts her glasses and kisses her on her cheek.
helen was safe and everyone is different key to unlock her.