sierransha08
01.07.2019 •
Chemistry
What is true about 1.0 mol ca and 1.0 mol mg they are equal in mass they contain the same number of atoms they have the same atomic mass their molar masses are equal
Solved
Show answers
More tips
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- C Computers and Internet How to Set Up Internet on iPhone? Detailed Guide with Step-by-Step Instructions...
- P Philosophy 8 привычек, чтобы достичь счастливой жизни...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- H Health and Medicine How to Choose the Right Glasses?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
- H Health and Medicine Flu: How to Recognize It by the First Symptoms?...
Answers on questions: Chemistry
- M Mathematics Isosceles triangle PQR is shown in the diagram Which statement about the medians of PQR is true? A. The medians of PQR are concurrent at a point and the medians of the...
- M Mathematics What is the slope of the line represented by the equation f(x) = 3/2x - 4? A. -4 B. -3/2 C. 3/2 D. 4...
- S Social Studies Discouraged workers are: a. workers who have a part time job but want a full time job. b. workers who find their current jobs unfulfilling and are considering a job change....
- M Mathematics Is 8,469,576 divisible by 6?...
- G Geography Which of the following letters identifies a state that nicholas spykman would argue has the most power? a) a b) b c)e d)c e)d...

Ответ:
- They contain the same number of atoms
Explanations:- Calcium and magnesium are two different elements and different elements have different atomic masses or molar masses. To calculate the mass we multiply the moles by the molar mass of the element. Since we have 1.0 mol of each but the molar masses are different, their masses will also be different.
1.0 mol of an element has atoms equals to Avogadro number that is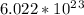 atoms. As we have 1.0 mol of each element, the number of atoms must also be same.
atoms. As we have 1.0 mol of each element, the number of atoms must also be same.
So, the only and only true statement is, "They contain the same number of atoms."
Ответ:
B) They contain the same number of atoms.
Ответ:
Sorry if it’s wrong i haven’t done that in a whole