What mass of aluminum can be plated onto an object in 755 minutes at 5.80 a of current? what mass of aluminum can be plated onto an object in 755 minutes at 5.80 a of current? 73.5 g 24.5 g 147 g 220. g 8.17 g?
Solved
Show answers
More tips
- H Health and Medicine What is the Normal Blood Sugar Level in a Healthy Person?...
- D Dating, Love, Relationships How Long Can Love Last?...
- A Auto and Moto Mastering One-Movement Parking: All You Need to Know...
- C Computers and Internet How to Properly Order Clothing from International Online Stores...
- H Health and Medicine Headache: A Comprehensive Guide to Treatment...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- A Auto and Moto Discovering the Leader: What is the Most Expensive Car in the World?...
- F Food and Cooking How to Quickly Put your Child to Sleep?...
- C Computers and Internet How to Create a Website for Free and Easy?...
- F Family and Home Parquet or laminate, which is better?...
Answers on questions: Chemistry
- C Chemistry Find the volume of 340 grams of BaS...
- E English HELPPP Which of the following is one of Aristotle s means of persuasion? Ο Α. The language of a speech O B. The speed of delivery O c. The purpose of a speech OD. The audience...
- H History Why did many Americans criticize TARP? -They believed TARP was helping businesses that deserved to fail. -They felt that TARP was only helping businesses that caused the crisis....
- E English Which sentence below shows correct use of an em dash? we’re hosting a party – you are invited, of course – later this month. we’re hosting a party you are invited – of course later...
- H History 2. why do you think the plague has been such a problem in the past? what effects has the plague had?...
- M Mathematics Can you please help me with this assignment. I will give you brainliest...

Ответ:
24.5 g
Explanation:
where Q= quantity of electricity in coloumbs
I = current in amperes = 5.80 A
t= time in seconds =755 min =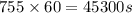
262740C of electricity deposits =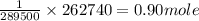 of Zn.
of Zn.
Mass of aluminium=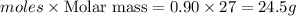
Ответ: