catalanb474
05.10.2021 •
Chemistry
What mass of C*O_{2} would be needed to weigh out to make 10.5 moles of carbon dioxide?(round answer to nearest tenth)
Solved
Show answers
More tips
- F Food and Cooking How to Make Chebureki: A Delicious Recipe for Russian Street Food...
- A Animals and plants Unraveling the Mystery of Loch Ness: What Does the Loch Ness Monster Look Like?...
- L Leisure and Entertainment Should You Buy a Ceramic Knife?...
- C Computers and Internet How to easily and quickly disable Firebug in Gmail and Google Docs...
- G Goods and services How to sew a ribbon: Tips for beginners...
- F Food and Cooking How to Make Mayonnaise at Home? Secrets of Homemade Mayonnaise...
- C Computers and Internet Which Phone is Best for Internet Surfing?...
- F Food and Cooking Everything You Need to Know About Pasta...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
Answers on questions: Chemistry
- C Chemistry (c) Assume you have an equilibrium mixture of [A], [B], and [C] at 298K and that the reaction is endothermic. i) What would happen to the concentration of C in this mixture if...
- C Chemistry 1. The directions for a laboratory activity call for 50 milliliters (ml) of solution A. A student accidentally takes 55 ml from the stock bottle. What should the student do with...
- C Chemistry The theory is connected to levels because the theory explains the pattern of activity of electrons around the nucleus of atoms...
- C Chemistry Fill in the best answer for each of the following: An ionic bond forms when atoms electrons. Another name for an ionic compound is a . Instead of forming molecules, ionic compounds...
- C Chemistry Choose the thermochemical equation that illustrates ΔH°f for K2SO4(s)....
- C Chemistry Use the standard reaction enthalpies given below to determine ΔH°rxn for the following reaction: P4(g) + 10 Cl2(g) → 4 PCl5(s) ΔH°rxn = ? Given: PCl5(s) → PCl3(g) + Cl2(g) ΔH°rxn...
- C Chemistry A reaction takes place between an acid and 0.5 gram of solid magnesium ribbon. Another reaction takes place between an acid and 0.5 grams of powdered magnesium which statement...
- H History What caused the British to impose taxes on the colonists in the first place? The high price of tea Expensive red uniforms The Intolerable Acts The cost of the French and Indian...
- M Mathematics Write the word sentence as an equation. Then solve. The sum of a number y and -3 is –8. Equation: Solution: y = 24...
- C Computers and Technology Some people worry that there won t be enough jobs in the future because computers will be able to do everything better than people can. Do you think that will happen? Why or...

Ответ:
10.5 moles of carbon dioxide?
462 g
Explanation:
Ответ:
The amount of heat required to convert H₂O to steam is : 382.62 kJ
Given data :
Mass of liquid water ( m ) = 150 g
Temperature of liquid water = 43.5°C
Temperature of steam = 130°C
Determine the amount of heat requiredThe amount of heat required = ∑ q1 + q2 + q3 ----- ( 1 )
where ;
q1 = heat required to change Temperature of water from 43.5°C to 100°C . q2 = heat required to change liquid water at 100°C to steam at 100°C
q3 = heat required to change temperature of steam at 100°C to 130°C
For q1M* S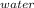 *ΔT
*ΔT
= 150 * 4.18 * ( 100 - 43.5 )
= 35425.5 J
For q2moles * ΔHvap
= (150 / 18 )* 40.67 * 1000
= 338916.67 J
For q3M * S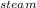 * ΔT
* ΔT
= 150 * 1.84 * ( 130 -100 )
= 8280 J
Back to equation ( 1 )
Amount of heat required = 35425.5 + 338916.67 + 8280 = 382622.17 J
≈ 382.62 kJ
Hence we can conclude that The amount of heat required to convert H₂O to steam is : 382.62 kJ.
Learn more about Specific heat of water :