What mass of potassium hypochlorite (fw-90.6 g/mol) must be added to 4.50 x 10 ml of water to give a solution with ph 10.20? [ka(hcio) 4.0 x 10-8] 0.032g ? 2.4 g 04.1 g 9.1 g 20. g
Solved
Show answers
More tips
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
Answers on questions: Chemistry
- C Chemistry Compound X has the formula C7H14. X reacts with one molar equivalent of hydrogen in the presence of a palladium catalyst to form 3-methylhexane. Treatment of X with ozone...
- C Chemistry Use the balanced combustion reaction above to calculate the enthalpy of combustion for C8H16. C8H16(1)= -174.5kJ/mol. I have no clue how to start this question and need...
- C Chemistry Calculate the pH at the equivalence point for titrating a 0.400 M solution of aniline, C6H5NH2, with 0.400 M HBr. (Hint: Volume is doubled at equivalence point)...
- C Chemistry If the particles formed through fission have less mass than the starting material, has the Law of Conservation of Energy been broken? A. Yes, nuclear reactions break atoms...
- C Chemistry Which of the choices is not a statement or direct application of the second law of thermodynamics? There are no 100% efficient heat engines. The change in internal energy...
- C Chemistry The balanced equation for the reaction of aqueous pb(clo3)2 with aqueous nai is pb(clo3)2(aq)+2nai(aq)⟶pbi2(s)+2naclo3(aq) what mass of precipitate will form if 1.50 l...
- C Chemistry If you have 560 grams of magnesium. what mass of magnesium oxide will be produced?...
- C Chemistry Salicylic acid, hoc6h4co2h, and its derivatives have been used as pain relievers for a long time. salicylic acid occurs in small amounts in the leaves, bark, and roots...
- C Chemistry Which of the following elements is the most reactive? O A. Fluorine (F) O B. Carbon (C) O O O O O C. Arsenic (As) O D. Sulfur (S) ....
- C Chemistry which of the following halides reacts more slowly in an SN1 reaction: 5-chloro-1,3-cyclopentadiene or 7-chloro-1,3,5-cycloheptatriene Group of answer choices 5-chloro-1,3-cyclopentadiene...

Ответ:
Answer : The mass of potassium hypochlorite is, 4.1 grams.
Explanation : Given,
pH = 10.20
Volume of water =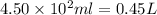
The decomposition of KClO will be :
Now the further reaction with water to give,
to give,
First we have to calculate the pOH.
Now we have to calculate the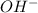 concentration.
concentration.
Now we have to calculate the base dissociation constant.
Formula used :
Now put all the given values in this formula, we get :
Now we have to calculate the concentration of .
.
The equilibrium constant expression of the reaction is:
As we know that,![[OH^-]=[HClO]=1.58\times 10^{-4}M](/tpl/images/0055/2981/ec220.png)
Now we have to calculate the moles of .
.
As we know that, the number of moles of are equal to the number of moles of KClO.
are equal to the number of moles of KClO.
So, the number of moles of KClO = 0.0449 mole
Now we have to calculate the mass of KClO.
Therefore, the mass of potassium hypochlorite is, 4.1 grams.
Ответ:
.
Step-by-step explanation: