What minimum mass of iron (II) nitrate must be added to 10.0 of a 0.0699 M phosphate solution in order to completely precipitate all of the phosphate as solid iron (II) phosphate? 2PO43–(aq) + 3Fe(NO3)2(aq) → Fe3(PO4)2(s) + 6NO3–(aq)
Solved
Show answers
More tips
- F Family and Home How to Choose the Perfect Air Conditioner for Your Life...
- H Health and Medicine Discover the Hidden Principles and Real Results of the Japanese Diet...
- H Health and Medicine Understanding Pregnancy Tests: What You Need to Know?...
- H Health and Medicine What Makes a Man a Man?...
- C Computers and Internet How to Get Rid of Spam in ICQ?...
- A Art and Culture Who Said The Less We Love a Woman, the More She Likes Us ?...
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
Answers on questions: Chemistry
- C Chemistry The ph of an solution shows the concentration of () ions....
- C Chemistry What effects can occur when heat energy is added to a system? select all that apply. a. increase in temperature b. decrease in temperature c. change of state d. phase change...
- C Chemistry What mass of compound must be weighed out to have a 0.0223 mol sample of h2c2o4 (m=90.04 g/mol)?...
- C Chemistry Where would the gravitational attraction between the earth and moon be the weakest...
- C Chemistry What is the molarity when 20 grams of lioh is added to 200 ml of water?...
- C Chemistry 5. water is a newtonian fluid, meaning that it has a constant viscosity no matter how much force you apply to it. how does the term non-newtonian colloidal mixture apply to oobleck?...
- C Chemistry QUESTION 1 What part of the brain is responsible for inhibiting our impulsive decisions and actions? O amydala O pituitary gland O the pre-frontal cortex O occiptial lobe...
- C Chemistry Calculate the amount of energy it will take to change 20.0 grams of Water from -3 degrees C to vapor (100 degrees C) at sea level. Show all your work. heat of fusion of water = 334...
- C Chemistry Can you think of another real world example that demonstrates air moving when it is heated?...
- C Chemistry The compound shown below is the product of a claisen condensation. draw structural formulas for the reactants: ester and enolate ion....

Ответ:
The minimum mass of iron (II) nitrate that must be added is 0.188 grams
Explanation:
To calculate the number of moles for given molarity, we use the equation:
Molarity of phosphate solution = 0.0699 M
Volume of solution = 10 mL = 0.010 L (Conversion factor: 1 L = 1000 mL)
Putting values in above equation, we get:
The given chemical equation follows:
By Stoichiometry of the reaction:
2 moles of phosphate solution reacts with 3 moles of iron (II) nitrate
So,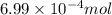 of phosphate solution will react with =
of phosphate solution will react with =  of iron (II) nitrate
of iron (II) nitrate
To calculate the number of moles, we use the equation:
Molar mass of iron (II) nitrate = 180 g/mol
Moles of iron (II) nitrate = moles
moles
Putting values in above equation, we get:
Hence, the minimum mass of iron (II) nitrate that must be added is 0.188 grams
Ответ:
the answer is 1.75 i just took the test