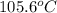What quantity in moles of potassium atoms do you have if you have 3.50 x 10^22 atoms of potassium.
Solved
Show answers
More tips
- F Family and Home How to Get Rid of Your Neighbors?...
- S Society and Politics How Could Nobody Know About the Dead Mountaineers?...
- H Health and Medicine How to Cure Adenoids?...
- H Health and Medicine Why Wearing a Back Brace Can Be Beneficial During Back Strain?...
- S Sport When and Where Will the 2014 World Cup be Held?...
- C Computers and Internet How to Choose a Monitor?...
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- C Computers and Internet How to Learn to Type Fast?...
Answers on questions: Chemistry
- C Chemistry What is the molar mass of C 10 H 16 ? Show math Hurry...
- C Chemistry Short out elements are compounds following: nitrogen, hydrogen, carbon dioxide, oxygen, Sodium Hydroxide, water, silver, ammonia....
- E English Type your response in the box. Now, read this essay. What do you think is the essay’s main purpose? Does it have only one purpose or are there others? Write down the purposes...
- M Mathematics A shipment of 12 television sets content to defective sets in how many ways can a hotel purchase five of these ads and receive at least two of the defective sets...
- H History Which two sentencing laws have contributed most to california s increasing lifer population...
- E English Statement that explains how the evidence supports and connects to the claim. A. Hook Sentence B. Explanation C. Transition D. Topic Sentence...

Ответ:
Answer : The volume of ethylene glycol is, 3.92 liters.
The boiling point of a solution is,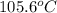
Explanation :
First we have to calculate the molality of ethylene glycol.
Formula used :
where,
i = Van't Hoff factor = 1 (for non-electrolyte)
m = molality of ethylene glycol = ?
Now put all the given values in the above formula, we get:
Now we have to calculate the mass of water.
Now we have to calculate the moles of ethylene glycol.
Moles of ethylene glycol = Molality × Mass of water
Moles of ethylene glycol = 10.8 mol/kg × 6.50kg = 70.2 mole
Now we have to calculate the mass of ethylene glycol.
Mass of ethylene glycol = Moles of ethylene glycol × Molar mass of ethylene glycol
Mass of ethylene glycol = 70.2 mole × 62.07 g/mole =4357.3 g
Now we have to calculate the volume of ethylene glycol.
The volume of ethylene glycol is, 3.92 liters.
Now we have to calculate the boiling point of solution.
Formula used for Elevation in boiling point :
where,
m = molality = 10.8 mol/kg
i = Van't Hoff factor = 1 (for non-electrolyte)
Now put all the given values in the above formula, we get the boiling point of a solution.
Therefore, the boiling point of a solution is,