dejaproctor17
22.12.2019 •
Chemistry
What's 4 ways to separate a mixture
Solved
Show answers
More tips
- A Art and Culture The History and Characteristics of Jazz Bands: A Deep Dive...
- F Food and Cooking From Latte to Espresso: Which Coffee Drink is the Most Popular on Earth?...
- C Computers and Internet How to Set Up Internet on iPhone? Detailed Guide with Step-by-Step Instructions...
- P Philosophy 8 привычек, чтобы достичь счастливой жизни...
- F Food and Cooking How Many Grams Are In a Tablespoon?...
- G Goods and services How to Choose the Right Iron: Purchase Tips...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- H Health and Medicine How to Choose the Right Glasses?...
- H Health and Medicine What vaccines do children need?...
- H Health and Medicine AKDS Vaccination: Ensure Your Child s Safety...
Answers on questions: Chemistry
- C Chemistry write a net ionic equation to explain the measured pH for any two solutions that have a pH less than 6 or greater than 8...
- C Chemistry Can you hypothesize different uses for sedimentary rock in our society?...
- C Chemistry Which best describes an enviromental adavntage of solar energy...
- C Chemistry Talk about something funny in your life that happend Mine- my grandpa was eating pizza and his tooth fell out while he was eating it...
- C Chemistry Why can calcium form a Ca2+ ion but not a Ca3+ ion...
- C Chemistry What mass of ammonia is formed when 3.58 moles of lithium nitride reacts with water? li3n + h2o à nh3 + li2o...
- C Chemistry Which three factor are most important in determining the composition of ocean water...
- E English Which best states how these two passages are different...
- M Mathematics Which series of transformations will not map figure h onto itself? (x + 0, y-2), reflection over y = 1 (x + 2, y - 0), reflection over x = 3 (x + 3, y + 3), reflection...
- E English 1. action-packed features have: (1 point) a lively tone but a slower pace a quiet tone and a slower pace a lively tone and a quicker pace a quiet tone but a quicker pace...

Ответ:
1.) Paper Chromatography
2.) Filtration
3.) Evaporation
4.) Simple Distillation
I concluded one more,
5.) Fractional Distillation
Ответ:
The amount of carbon sulfide prepared is 859.1724 grams.
Explanation:
To calculate the concentration of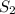 , we use the equation:
, we use the equation:
We are given:
Moles of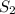 = 12.5 moles
= 12.5 moles
Volume of solution = 5.3 L
Putting values in above equation, we get:
For the reaction of sulfur and carbon, the equation follows:
At 2.36
2.36
At 2.36 - x x
2.36 - x x
The expression for equilibrium constant for the above reaction follows:We are given:
Putting values in above equation, we get:

Now, calculating the moles of carbon sulfide using equation 1, we get:Molarity of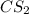 = 2.133 M
= 2.133 M
Volume of solution = 5.30 L
Putting values in equation 1, we get:

To calculate the mass of carbon sulfide, we use the equation:Moles of carbon sulfide = 11.3049 mol
Molar mass of carbon sulfide = 76 g/mol
Putting values in above equation, we get:
Hence, the amount of carbon sulfide prepared is 859.1724 grams.